Vol. 19 - Num. 74
Originales
Uso del test rápido de detección de antígeno estreptocócico en la consulta de Atención Primaria
Silvia Burgaya Subiranaa, Mireya Cabral Salvadoresa, Anna M.ª Bonet Esteveb, Esperança Macià Rieradevalla, Anna M.ª Ramos Calvoc
aPediatra. ABS Manlleu. Manlleu. Barcelona. España.
bUnidad de Farmacia. Gerencia Territorial de la Cataluña Central. Barcelona. España.
cMédico de familia. ABS Manlleu. Manlleu. Barcelona. España.
Correspondencia: S Burgaya. Correo electrónico: sburgaya.cc.ics@gencat.cat
Cómo citar este artículo: Burgaya Subirana S, Cabral Salvadores M, Bonet Esteve AM, Macià Rieradevall E, Ramos Calvo AM. Uso del test rápido de detección de antígeno estreptocócico en la consulta de Atención Primaria. Rev Pediatr Aten Primaria. 2017;19:119-25.
Publicado en Internet: 07-06-2017 - Número de visitas: 44080
Resumen
Introducción: la faringoamigdalitis aguda por Streptococcus pyogenes es uno de los diagnósticos más frecuentes en Pediatría. Para identificarla clínicamente se han propuesto varias escalas de predicción clínica (Centor y Centor modificada por McIsaac), pero para el diagnóstico etiológico se recomienda el uso del test rápido de detección antigénica de estreptococo. Este test tiene una alta sensibilidad y especificidad y es de fácil aplicación. Nuestro estudio tiene como objetivo conocer el uso de este test en la consulta de Pediatría de Atención Primaria de nuestra área de influencia.
Material y métodos: se ha realizado un estudio observacional retrospectivo de todos los pacientes de las comarcas centrales de Cataluña con diagnóstico de faringoamigdalitis, faringitis aguda, amigdalitis estreptocócica y amigdalitis aguda desde el 1 de enero de 2015 al 31 de enero de 2016.
Resultados: se han estudiado 12 678 episodios de faringoamigdalitis aguda estreptocócica en 10 276 pacientes. Se realizaron 1274 test rápido de detección (10,05%). El 47,02% resultó positivo. La escala de Centor modificada se registró en 1240 pacientes (9,78%); 6933 pacientes (53,81%) recibieron tratamiento antibiótico. Al 89,67% de estos no se les había realizado test rápido de detección. La amoxicilina fue el antibiótico más usado.
Conclusiones: constatamos una baja utilización del test rápido de detección y un bajo registro de la escala de Centor. Se ha observado un uso excesivo de antibióticos, pero los antibióticos utilizados se adaptan a las recomendaciones de las guías terapéuticas.
Palabras clave
● Faringitis ● Técnicas bacteriológicas ● Test diagnóstico ● TonsilitisINTRODUCCIÓN
La faringoamigdalitis aguda (FAA) es un proceso inflamatorio de la mucosa y las estructuras del área faringoamigdalar. Clínicamente se caracteriza por fiebre, odinofagia e hipertrofia amigdalar acompañada de eritema, edema, exudado, úlceras o vesículas.
La FAA estreptocócica es uno de los diagnósticos más frecuentes en la consulta de Pediatría de Atención Primaria (PAP) y es la primera causa de utilización de antibióticos en todo el mundo. A pesar de esto, la mayoría de las FAA son víricas y solo se consideran de etiología estreptocócica alrededor del 15% del total de los diagnósticos en niños, pudiendo aumentar hasta el 30% en periodos epidémicos1. La importancia de la identificación del Streptococcus pyogenes como agente etiológico de una FAA radica en las posibles complicaciones que puede comportar: complicaciones supurativas (1-2%) como otitis media, sinusitis aguda, absceso periamigdalino, mastoiditis o absceso retrofaríngeo, y complicaciones no supurativas como fiebre reumática, glomerulonefritis posestreptocócica o artritis reactiva. La utilización de antibióticos para tratar la FAA estreptocócica y evitar estas complicaciones se debate en la actualidad. En una revisión de las diferentes guías de práctica clínica para su manejo, realizada por García Vera et al.2 para el Grupo de Patología Infecciosa de la Asociación Española de Pediatría de Atención Primaria (AEPap), se observó que dos de estas guías (NICE, SIGN)3,4 consideran innecesario tratar las FAA estreptocócicas, salvo que produzcan una afectación importante del estado del paciente, mientras que el resto prefieren hacer un diagnóstico etiológico y tratarlas con antimicrobianos si se identifican como FAA estreptocócica1,5-9.
La clínica y los datos de la exploración física aportan poca información para poder diferenciar la FAA estreptocócica del resto, ya que ningún síntoma aislado ofrece un cociente de probabilidad positivo para superar un 50% de probabilidad de FAA estreptocócica. Se han elaborado escalas de predicción clínica (Centor y Centor modificada por McIsaac) (Tabla 1), pero estas ofrecen una aproximación máxima al diagnóstico de un 62%. Por tanto, estas escalas deberían servir para seleccionar los pacientes candidatos a aplicar las pruebas diagnósticas y no para realizar un diagnóstico etiológico2,10-14.

El gold standard para el diagnóstico de FAA estreptocócica es el cultivo faríngeo. Tiene una sensibilidad del 90-95% y una especificidad del 99%, pero tiene el inconveniente de que su resultado se demora al menos 48 horas. Desde los años 80 se han ido desarrollando test rápidos de diagnóstico (TRD) antigénico basados en la detección del antígeno carbohidrato específico de la pared celular del Streptoccocus pyogenes. Inicialmente estos test ofrecían una alta sensibilidad y una baja especificidad, pero los últimos años se han ido desarrollando técnicas basadas en enizimoinmunoanálisis e immunoanálisis óptico, con una precisión diagnóstica similar al cultivo (99% de sensibilidad y 95-99% de especificidad). Estas técnicas tienen la ventaja de ofrecer resultados en 5-10 minutos a un bajo coste (2,67 euros/test)2,11-13.
Además, estudios recientes han demostrado que el empleo del TRD reduce el uso de antibióticos hasta un 42%11-13.
Desde hace dos años, en las consultas de PAP de los centros gestionados por el Institut Català de la Salut se dispone de TRD para el diagnóstico etiológico de la FAA estreptocócica. El objetivo de nuestro estudio es conocer el uso del test después de su implementación de forma generalizada en las consultas de PAP de nuestra área de influencia.
MATERIAL Y MÉTODOS
Estudio observacional retrospectivo sobre una base poblacional de 64 172 niños, todos ellos en edades comprendidas entre 0 y 14 años procedentes de los 32 centros de salud gestionados por el Institut Català de la Salut en la Cataluña Central.
Se incluyeron en el análisis todos los episodios diagnósticos de faringoamigdalitis, faringitis aguda, amigdalitis aguda y amigdalitis estreptocócica de la historia clínica electrónica durante el periodo desde el 1 de enero de 2015 al 31 de enero de 2016.
Se analizaron las variables de edad, tipo y momento del diagnóstico, escala de Centor modificada, número de TRD realizados y su resultado en función de la escala y prescripción de antibióticos según resultados del test y de la escala de Centor.
Se utilizó la media y la desviación estándar para la descripción de las variables cuantitativas con distribución normal y tasas/porcentajes para las dicotómicas/ordinales. Las diferencias de resultados se calcularon mediante odds ratio (OR), con un intervalo de confianza del 95% y un grado de significación estadística de p < 0,05, utilizando el test de χ2.
Para el análisis estadístico de los datos se utilizaron los programas SPSS® versión 18 y Epi Info® versión 7.
RESULTADOS
Se registraron 12 678 diagnósticos de faringoamigdalitis, faringitis aguda, amigdalitis aguda o amigdalitis estreptocócica en 10 276 pacientes durante el periodo de estudio. La edad media de los pacientes estudiados fue de 6 años y 11 meses (desviación estándar [DE] +/- 3 años y 8 meses). Se realizaron 1274 TRD (10,05%), de los cuales 599 (47,02%) resultaron positivos; 101 (7,93%) TDR no tenían indicado el resultado. La escala de Centor modificada se registró en 1240 episodios (9,78%). La puntuación más registrada fue de 3 (38,39%). El mes con más registros diagnósticos fue noviembre (11,63%). El 21,29% de los episodios de FAA se registraron en menores de tres años.
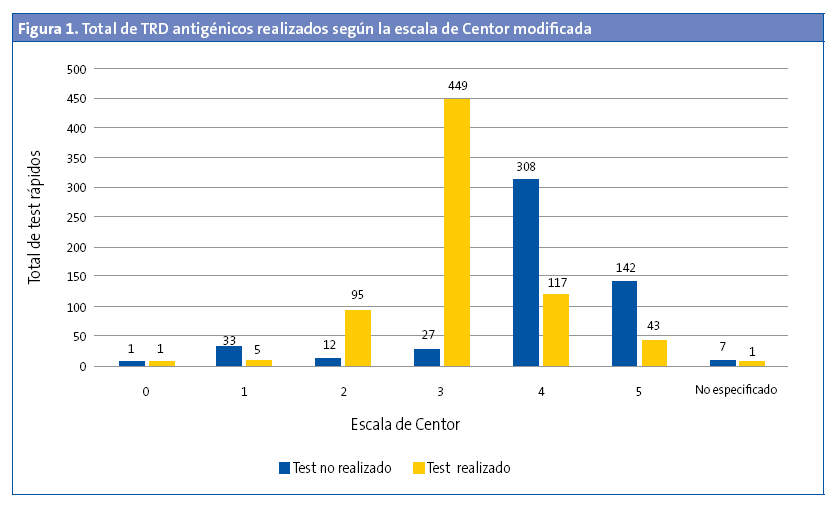
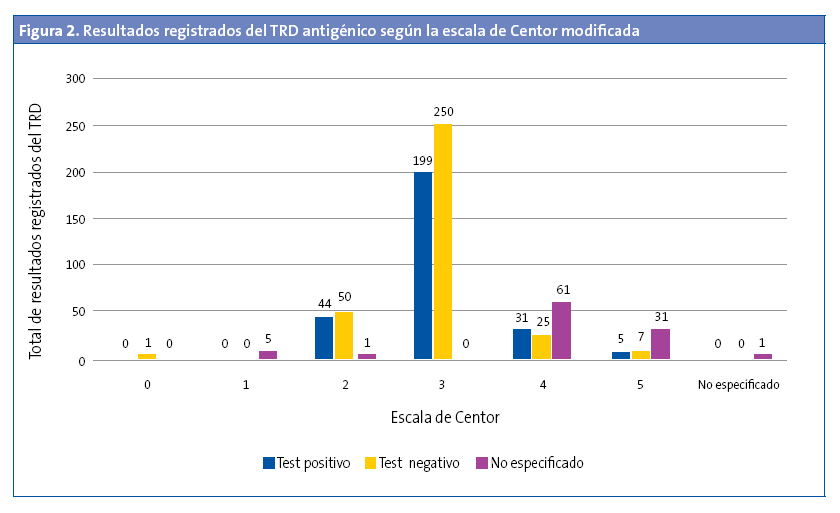
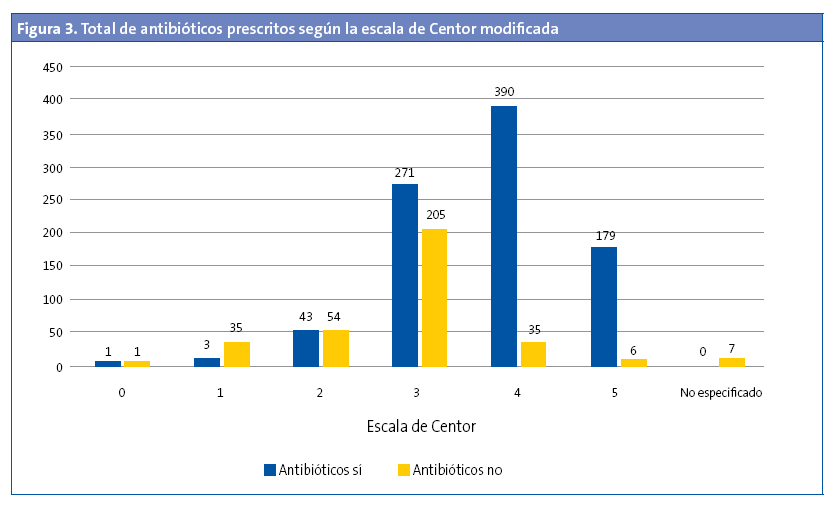
Se realizó TRD en el 5,30% de los menores de tres años (OR: 0,45 [0,37-0,54]; p < 0,05). De estos, el 27,27% tenían un TRD positivo. En 529 (42,66%) de los episodios se registró la escala de Centor modificada, pero no se realizó el test (Fig. 1). No se realizó en el 43,92% (477 de 1086) de los que tenían una puntuación de 3, 4 y 5. Al 94,33% de los pacientes con una puntuación de 3 se les había solicitado el TRD (OR: 31,9 [21,0-48,3]; p < 0,001), pero solo se solicitó TRD en el 26,22% de los pacientes que tenían una puntuación de 4 y 5. El 57,5% de los pacientes con una puntuación 4 y 5 tenían codificado como resultado del test “no procede realizar test” (Fig. 2). De todos los pacientes con una puntuación 4 y 5, el 47,05% tenía un TRD negativo (OR: 0,71 [0,43-1,19]; p > 0,05). Se administró antibiótico en 6933 (54,69%) episodios. En un 89,67% de estos episodios no se realizó previamente el TRD. El 95,69% de episodios en los que se prescribió antibiótico sin realizar un TDR tenía una puntuación de Centor 4 o 5. En la Fig. 3 se muestra la distribución de la prescripción de antibióticos según el resultado de la escala de Centor. En 34 (5,92%) de los episodios con un test negativo se prescribió antibiótico. Los antibióticos más utilizados para el tratamiento fueron las penicilinas, en un 93,72% de los casos. La penicilina más usada fue amoxicilina (84,22%). En el 95,1% de los casos se prescribió antibiótico en las primeras 24 horas.
DISCUSIÓN
Actualmente, existen muchos estudios realizados a nivel hospitalario sobre el diagnóstico y manejo de las FAA estreptocócica, pero hay pocos estudios realizados a nivel de Atención Primaria (AP). En nuestro estudio se observa que después de la implementación generalizada del TRD en la consulta de PAP, en nuestra área de influencia se hace un uso escaso del mismo (solo se ha realizado en un 10,05% de los diagnósticos). Seguramente este hecho se debe a que los pediatras de nuestra área siguen utilizando criterios clínicos y no etiológicos para el diagnóstico de la FAA estreptocócica. La edad media del diagnóstico (6 años y 11 meses) concuerda con la bibliografía, así como la predominancia de FAA de origen vírico en menores de tres años (el 27,3% de los test realizados en menores de tres años resultó positivo)1-14. Por esta misma razón, solo se ha realizado el TRD en el 5,3% de los menores de esta edad. Realizar el test a todos los menores de tres años no sería coste-efectivo, si ya de entrada la sospecha diagnóstica es de FAA vírica.
Se ha observado un bajo porcentaje de registros de la escala de Centor modificada (9,78%). Podría deberse a la gran presión asistencial a que están sometidos los pediatras de AP y que provocaría que, aunque realizaran la escala, obviasen apuntarlo para poder atender a los pacientes de forma más rápida. Otra causa que podría justificar esta observación podría ser que, tal y como hemos dicho anteriormente, los pediatras usarían su criterio clínico directamente sin utilizar ninguna escala para la aproximación diagnóstica. En todo caso, este aspecto podría ser objeto de estudio en trabajos posteriores.
Aproximadamente la mitad de los TRD realizados resultaron positivos. En los episodios con una puntuación 4 y 5 también fueron positivos la mitad de los TRD, sin observar diferencias significativas al comparar el resultado del test (positivo/negativo) y la puntuación de Centor modificada. Se resaltan estos datos porque lo que reflejan es que, a pesar de que se han diseñado escalas para la aproximación diagnóstica, estas son, tal y como indica la mayor parte de la bibliografía, una aproximación y no sirven para un diagnóstico etiológico seguro2,6,7,9-11,13-15. La bibliografía baraja una aproximación máxima del 62%2. Diversos estudios de coste-efectividad demuestran que realizar el TRD a todos los pacientes con una puntuación ≥ 3 es la opción más coste-efectiva11,16,17. A pesar de estos datos, en nuestro estudio hemos detectado que en un 43,92% de los episodios con puntuación de 3, 4 y 5 no se les realizó el TRD y en un 95,69% de episodios en los que se prescribió antibiótico sin realizar un TDRA tenía una puntuación de Centor 4 o 5, por lo que se deduce que con una puntuación de 4 y 5 se prescribió antibiótico directamente, sin realizar el TRD en la mayoría de los casos y, por tanto, utilizando la escala de valoración clínica para la decisión final de tratamiento. El 57,5% de los registros con una puntuación de Centor modificada de 4 y 5 no tenían resultado del test y se les había codificado como resultado: “no procede la realización del test”. Esto nos indica que algunos pediatras aún no tienen claro que con puntuaciones altas de la escala de Centor modificada es necesario realizar el test.
Hemos observado un alto porcentaje de prescripción de antibióticos. El 54,69% de los pacientes diagnosticados de FAA habían recibido antibiótico y la mayoría (95,1%) lo había recibido en las primeras 24 horas. La mayor parte de esta prescripción había sido empírica, ya que al 89,67% de estos no se les había realizado el TRD. Los antibióticos utilizados para el tratamiento de la FAS se adaptan a las guías de práctica clínica en la mayoría de los casos.
CONCLUSIONES
Es preciso concienciar a los pediatras sobre la conveniencia de la realización del TRD ante una clínica compatible de faringoamidalitis. Se observa un bajo uso de estos test diagnósticos. Está ampliamente demostrado que las escalas de aproximación diagnóstica y la clínica, valorada incluso por un pediatra experimentado, tienen una correlación muy pobre con la confirmación microbiológica. Con el empleo del TRD se pretende realizar un correcto diagnóstico y disminuir el uso indiscriminado de antibióticos y el gasto sanitario.
Ante los resultados del presente estudio y la bibliografía aportada, creemos que se debe iniciar una campaña activa de concienciación de los pediatras sobre el uso del TRD en los centros de AP de nuestra área de influencia y realizar estudios posteriores para confirmar la buena implementación del test.
CONFLICTO DE INTERESES
Los autores declaran no presentar conflictos de intereses en relación con la preparación y publicación de este artículo.
ABREVIATURAS: AEPap: Asociación Española de Pediatría de Atención Primaria · AP: Atención Primaria · DE: desviación estándar · FAA: faringoamigdalitis aguda · OR: odds ratio · PAP: Pediatría de Atención Primaria · TRD: test rápido de diagnóstico.
BIBLIOGRAFÍA
- De la Flor i Brú J. Infecciones de vías respiratorias altas-1: faringitis aguda y recurrente. Pediatr Integral. 2013;17:241-61.
- García Vera C; Grupo de Patología Infecciosa de la Asociación Española de Pediatría de Atención Primaria. Utilidad del test rápido de detección de antígeno estreptocócico (TRDA) en el abordaje de la faringoamigdalitis aguda en Pediatría. En: AEPap [en línea] [consultado el 31/05/2017]. Disponible en www.aepap.org/sites/default/files/gpi_utilidad_trda_estreptoccico.pdf
- NICE Clinical Guideline 69. Respiratory tract infections-antibiotic prescribing. Prescribing of antibiotics for self-limiting respiratory tract infections in adults and children in primary care. Londres: NICE; 2008.
- Management of sore throat and indications for tonsillectomy. A national clinical guideline. En: SIGN Scottish Intercollegiate Guidelines Network [en línea] [consultado el 31/05/2017]. Disponible en www.sign.ac.uk/pdf/sign117.pdf
- Clinical Practice Guideline working group. Guideline for the diagnosis and management of acute pharyngitis. En: Alberta Medical Association [en línea] [consultado el 31/05/2017]. Disponible en www.topalbertadoctors.org/download/368/acute_pharyngitis_guideline.pdf.
- Bercedo Sanz A, Cortés Rico O, García Vera C, Montón Álvarez JL. Normas de calidad para el diagnóstico y tratamiento de la faringoamigdalitis aguda en Pediatría de Atención Primaria. Protocolos de GVR (publicación P-GVR-10). En: Respirar [en línea] [consultado el 31/05/2017]. Disponible en www.respirar.org/images/pdf/grupovias/faringoamigdalitis2011.pdf
- Anjos LM, Marcondes MB, Lima MF, Mondelli AL, Okoshi MP. Streptococcal acute pharingitis. Rev Soc Bras Med Trop. 2014;47:409-13.
- Picazo JJ, Pérez-Cecilia E, Herreras A; Grupo DIRA en Atención Primaria Estudio de las infecciones respiratorias extrahospitalarias. Estudio DIRA. Enferm Infecc Microbiol Clin. 2003;21:410-6.
- Danchin MH, Rogers S, Kelpie L, Selvaraj G, Curtis N, Carlin JB, et al. Burden of acute sore throat and group A streptococcal pharyngitis in scholl-aged children and their families in Australia. Pediatrics. 2007;120:950-7.
- Roggen I, van Berlaer G, Gordts F, Pierard D, Hubloue I. Centor criteria in children in a paediatric emergency department: for what it is worth. BMJ Open. 2013;3. pii: e002712.
- Orda U, Mitra B, Ordo S, Fitzgerald M, Gunnausson R, Rofe G, et al. Point of care testing for group A streptococci in patients presenting with pharyngitis will improve appropriate antibiotic prescription. Emerg Med Australas. 2016;28:199-204.
- De la For J, Parellada N. Utilització sistemàtica de tests de diagnòstic ràpid en una consulta de Pediatría d’atenció primària. Pediatr Catalana. 2009;69:75-84.
- Kose E, Sirin Kose S, Akca D, Yildiz K, Elmas C, Baris M, et al. The effect of rapid antigen detection test on antibiotic prescription decision of clinicians and reducing antibiotic costs in children with acute pharyngitis. J Trop Pediatr. 2016;62:308-15.
- Mazur E, Bochynska E, Juda M, Kozioll-Montewka M. Empirical validation of Polich guidelines for the management of acute streptococcal pharyngitis in children. Int J Pediatr Otorrhinolaryngol. 2014;8:102-6.
- McIsaac WJ, Kellner JD, Aufricht P, Vanjaka A, Low DE. Empirical validation of guidelines for the management of pharyngitis in children and adults. JAMA. 2004;291:1587-95.
- Giráldez-Garcia C, Rubio B, Gallegos-Braun JF, Imaz I, González-Enríquez J, Sarriá-Santamera A. Diagnosis and management of acute pharyngitis in a paediatric population: a cost-effectiveness analysis. Eur J Pediatr. 2011;170:1059-67.
- Van Howe RS, Kusnier LP. Diagnosis and management of pharyngitis in a pediatric population based on cost-effectiveness and projected health outcomes. Pediatrics. 2006;117:609-19.




