Vol. 20 - Num. 80
Originales
Perfil nutricional y contenido de azúcares de los preparados lácteos para niños pequeños disponibles en los supermercados
Ana Frades Payoa, Miguel Ángel Royo Bordonadab
aVeterinaria de Equipo de Atención Primaria. Servicio Extremeño de Salud. Badajoz. España.
bInstituto de Salud Carlos III. Madrid. España.
Correspondencia: A Frades. Correo electrónico: anafradesp@gmail.com
Cómo citar este artículo: Frades Payo A, Royo Bordonada MA. Perfil nutricional y contenido de azúcares de los preparados lácteos para niños pequeños disponibles en los supermercados. Rev Pediatr Aten Primaria. 2018;20:353-63.
Publicado en Internet: 27-11-2018 - Número de visitas: 42191
Resumen
Objetivos: analizar el perfil nutricional, el contenido de azúcares libres y la presencia de alegaciones de salud o avales científico-sanitarios de las fórmulas lácteas de crecimiento para niños de uno a tres años. El perfil nutricional se comparará con el de la leche entera de vaca.
Material y métodos: estudio descriptivo de una muestra de 20 tipos distintos de leches de crecimiento comercializadas en los supermercados de Badajoz (España). La información nutricional y sobre alegaciones o avales se obtuvo del etiquetado de los productos. La cantidad de azúcares libres se estimó mediante la diferencia entre los azúcares totales y el contenido en azúcares de la leche de vaca.
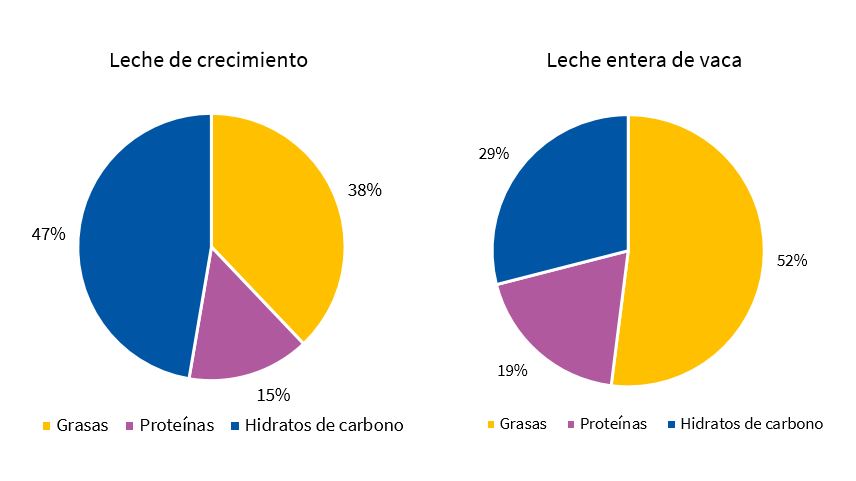
Resultados: comparadas con la leche entera de vaca, las leches de crecimiento aportan más calorías (67,7 frente a 65 kcal/100 ml) e hidratos de carbono (el 47,5 frente al 29%), y menos grasas (el 37,7 frente al 52%) y proteínas (el 14,8 frente al 19%). Los azúcares libres aportaron entre el 3 y el 22% del contenido calórico total. Todos los productos presentaron algún tipo de alegación y el 60% hicieron uso de alegaciones de salud no autorizadas y contaron con el aval de la Asociación Española de Pediatría.
Conclusiones: el alto contenido en azúcares añadidos de las leches de crecimiento es incompatible con las recomendaciones de la Organización Mundial de la Salud y de los expertos en nutrición. Por tanto, debería regularse su composición nutricional, con un etiquetado más informativo, de tipo interpretativo, permitiendo el uso de alegaciones o avales científicos únicamente en productos que sigan tales recomendaciones.
Palabras clave
● Marketing ● Azúcar ● Etiquetado ● Leches de crecimiento ● Niños ● ObesidadINTRODUCCIÓN
En España, aproximadamente uno de cada tres niños presenta sobrepeso u obesidad, una de las cifras más altas de Europa1. Aunque la prevalencia de obesidad infantil parece estabilizada en los últimos años, el porcentaje de padres que tienen una percepción errónea del exceso de peso de sus hijos sigue creciendo de forma alarmante2. Entre los diversos factores que contribuyen a esta epidemia de obesidad infantil, las bebidas azucaradas se han asociado a un mayor riesgo desde la infancia precoz3. Además, el consumo excesivo de azúcares añadidos en niños también se asocia con la aparición de caries dental y de varios factores de riesgo cardiovascular, como presión arterial elevada, hipercolesterolemia y diabetes mellitus tipo 24.
El consumo de productos azucarados a edades tempranas puede incrementar la preferencia por el azúcar en los alimentos, la cual se mantiene hasta la adolescencia, influyendo así en la adquisición y mantenimiento de hábitos alimentarios poco saludables5. En estudios experimentales con roedores se ha determinado que el consumo de azúcar activa un mecanismo de retroalimentación metabólica en el circuito de la recompensa del cerebro6. De forma similar, algunas personas, bajo situaciones de estrés, serían susceptibles de engancharse al azúcar, por activación del circuito cerebral de la recompensa y, en consecuencia, serían más vulnerables a la obesidad y demás patologías relacionadas con el consumo de azúcar6.
La Organización Mundial de la Salud (OMS) recomienda la lactancia materna exclusiva desde la primera hora de vida hasta los 6 meses, con posterior introducción progresiva de alimentos complementarios, variados y nutritivos, sin añadir sal o azúcar a los mismos y sin abandonar la lactancia materna7,8. Además, aconseja limitar el consumo de azúcares libres como máximo al 10% de la ingesta calórica y señala que reducirlos por debajo del 5% ofrecería beneficios adicionales, especialmente en la prevención de la caries dental9-12. Los comités de nutrición de The European Society for Paediatric Gastroenterology Hepatology and Nutrition y de la Asociación Española de Pediatría Extrahospitalaria y Atención Primaria recomiendan el consumo de leche entera en menores de 2 años y 2-3 raciones de lácteos para niños en edad preescolar y escolar13,14.
En los últimos años, en el mercado han aparecido preparados lácteos para niños de corta edad (1-3 años), un tipo de bebidas elaboradas a base de leche de vaca y enriquecidas con diversos micronutrientes, que se comercializan bajo las denominaciones “leche de crecimiento”, “alimento lácteo infantil” o “bebida láctea infantil”, que no están reguladas de forma específica por la legislación de la Unión Europea. Pese a las recomendaciones de la OMS y de los comités de expertos en nutrición, y al contrario de lo que sucede con las fórmulas lácteas de inicio y de continuación, estas bebidas, que en lo sucesivo denominaremos genéricamente como “leches de crecimiento” (LC), suelen contener cantidades variables de azúcares añadidos15.
Objetivos
Analizar el perfil nutricional y el contenido de azúcares libres de las LC comercializadas para niños de 1 a 3 años, y compararlos con los de la leche entera de vaca. Asimismo, se valorará la presencia de alegaciones nutricionales o de salud y de avales científicos o sanitarios.
MATERIAL Y MÉTODOS
Diseño y muestra del estudio
Estudio descriptivo del contenido de azúcares en las LC dirigidas a niños pequeños (de uno a tres o más años) en los supermercados de la ciudad de Badajoz (España). Para obtener la muestra del estudio, se visitaron en marzo de 2017 los establecimientos de Badajoz asociados a grandes cadenas de distribución con implantación a nivel nacional (Hipercor®, Carrefour®, Mercadona®, Aldi®, LIDL®, Supermercados El Corte Inglés®, Carrefour® Express y DIA®). En cada centro se revisaron los lineales de alimentación infantil y los de productos lácteos, encontrándose 20 variedades distintas de LC de seis marcas comerciales (Puleva®, Nestlé®, Hacendado®, Celta®, Kaiku® y DIA®) destinadas a niños de uno a tres años, que constituyen la muestra del estudio.
Recogida de datos y variables del estudio
Los datos sobre el contenido calórico y de azúcares totales y añadidos de cada producto seleccionado se obtuvieron de la información que facilita el fabricante en las etiquetas de los envases disponibles a la venta. La OMS define como azúcares libres a los monosacáridos y los disacáridos añadidos a los alimentos y las bebidas por el fabricante, el cocinero o el consumidor (azúcares añadidos), más los azúcares naturalmente presentes en la miel, los jarabes, los zumos de frutas y los concentrados de zumosde frutas16. En esta definición de azúcares libres no incluye los azúcares que, pese a no haber sido añadidos, se hayan liberado por un proceso tecnológico17. Para detectar la presencia de azúcares añadidos se revisó la lista de ingredientes, recogiéndose su denominación en cada caso. La cantidad total de azúcares por 100 ml de producto se obtuvo de la tabla de composición nutricional. Como las tablas no proporcionan la cantidad de azúcares añadidos, para estimar la cantidad de azúcares libres (los no contenidos de forma natural en la leche), se calculó la diferencia entre los azúcares totales y el contenido en azúcares de la leche de vaca desnatada o semidesnatada (4,6 g/100 g), que fueron las utilizadas como ingredientes de las LC, a partir de la información de la Base de Datos Española de Composición de Alimentos (BEDCA) (www.bedca.net). Puesto que se desconoce el porcentaje de leche que contiene cada producto (dato no proporcionado en la etiqueta), el estimador de azúcares libres es conservador y debe considerarse como el límite inferior en caso de que los productos únicamente contuviesen dos ingredientes: leche de vaca y azúcares añadidos.
Se registró la presencia de declaraciones nutricionales o de propiedades saludables y avales sanitarios o científicos en el envase de los productos y en los envoltorios o embalajes de productos comercializados en grupos de varias unidades. El Reglamento 1924/2006 define declaración de propiedades saludables como cualquier mensaje o representación que afirme, sugiera o dé a entender que existe una relación entre una categoría de alimentos, un alimento o uno de sus constituyentes, y la salud; actualmente existen 261 declaraciones de salud autorizadas. También se recogió información del precio de venta al público y la ubicación de los productos dentro de cada establecimiento.
Análisis estadístico
A partir del listado de ingredientes del etiquetado de cada producto, se calculó el porcentaje de FLC con azúcares añadidos. Asimismo, se calculó la media de azúcares totales y libres. En el cálculo de la media de azúcares libres no se tuvieron en cuenta los productos que no declaran azúcares añadidos en la lista de ingredientes, ni los que, declarando azúcares añadidos, su contenido en azúcares totales fue inferior al de la leche de vaca.
Para calcular el porcentaje calórico de cada producto aportado por los azúcares totales y los azúcares libres, considerando que un gramo de azúcar aporta 4 kcal, se multiplicó esta cifra por los gramos de azúcares contenidos en 100 ml de producto y se dividió el resultado por las kcal totales. El mismo procedimiento se utilizó para calcular el aporte calórico de grasas (9 kcal/g) y proteínas (4 kcal/g).
Finalmente, se cuantificó el número y porcentaje de productos con presencia de alegaciones nutricionales o de salud y avales sanitarios o científicos en el etiquetado o envase de estos.
RESULTADOS
El 70% (14/20) de las LC están elaboradas a base de leche semidesnatada y el resto a base de leche desnatada. El aporte energético medio de las LC estudiadas es de 67,7 kcal/100 ml, ligeramente superior al de la leche entera de vaca (65 kcal/100 ml) y entre 1,5 y 2 veces mayor que las leches semidesnatada y desnatada con que están elaborados estos productos. Las cantidades medias de grasas, proteínas e hidratos de carbono presentes en las LC son de 2,8, 2,5 y 8 g/100 ml de producto, lo cual equivale a un 37,7, 14,8 y 47,5% del aporte calórico total del producto respectivamente. Por tanto, las LC presentan un contenido alto de azúcares y bajo de proteínas y grasas en comparación con la leche entera de vaca, recomendada para este grupo de edad (Fig. 1).
| Figura 1. Perfil nutricional de la leche entera de vaca y las leches de crecimiento disponibles en los supermercados de Badajoz (2017) |
|---|
 |

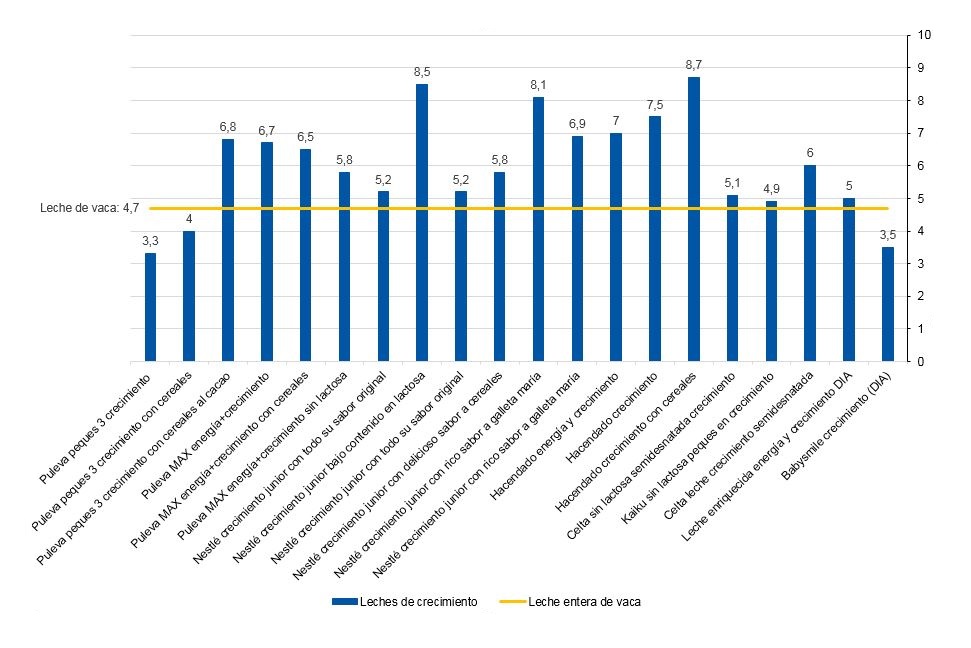
De las 20 LC analizadas, 17 (85%) contienen azúcares añadidos en su lista de ingredientes bajo una o varias de las siguientes denominaciones: azúcar, fructosa, sacarosa, maltodextrina, miel y caramelo (Tabla 1). La media de azúcares totales es de 6 g/100 ml de producto y la de azúcares libres de 2,1 g/100 ml. De las 20 presentaciones comerciales, 17 (85%) superaron el contenido en azúcares totales de la leche entera de vaca (4,7 g/100 ml), con cifras que variaron entre los 4,9 y 8,7 g/100 ml (Fig. 2). Sin embargo, tres de estos productos no declararon la presencia de azúcares añadidos en la lista de ingredientes, destacando la LC semidesnatada Celta con 6 g de azúcar/100 ml (Tabla 1). Además, se constató la existencia de tres productos que contenían menos cantidad de azúcares totales que la leche semidesnatada con que estaban elaborados, pese a que declaraban la presencia de azúcares añadidos en la lista de ingredientes (Tabla 1).
| Tabla 1. Contenido en azúcares de las leches de crecimiento disponibles en los supermercados de Badajoz (2017) | |||||
|---|---|---|---|---|---|
| Presentación comercial | Tipo de leche | Edad | Azúcares añadidos* | Azúcares totales g/100 ml |
Azúcares libres g/100 ml** |
| Puleva peques 3 crecimiento | Semidesnatada | +12 meses | Azúcar, maltodextrina, fructosa | 3,3 | |
| Puleva peques 3 crecimiento con cereales | Semidesnatada | +12 meses | Sacarosa, maltodextrina, fructosa | 4 | |
| Puleva peques 3 crecimiento con cereales al cacao | Semidesnatada | +12 meses | Sacarosa, maltodextrinas, fructosa | 6,8 | 2,2 |
| Puleva MAX energía + crecimiento | Semidesnatada | +3 años | Azúcar, fructosa | 6,7 | 2,1 |
| Puleva MAX energía + crecimiento con cereales | Semidesnatada | +3 años | Azúcar, fructosa | 6,5 | 1,9 |
| Puleva MAX energía + crecimiento sin lactosa | Semidesnatada | +3 años | Azúcar | 5,8 | 1,2 |
| Nestlé crecimiento junior con todo su sabor original | Desnatada | +12 meses | Dextrinomaltosa, azúcar, lactosa | 5,2 | 0,6 |
| Nestlé crecimiento junior bajo contenido en lactosa | Desnatada | +12 meses | Dextrinomaltosa, azúcar | 8,5 | 3,9 |
| Nestlé crecimiento junior con todo su sabor original | Desnatada | +2 años | Dextrinomaltosa, azúcar, lactosa | 5,2 | 0,6 |
| Nestlé crecimiento junior con delicioso sabor a cereales | Desnatada | +12 meses | Dextrinomaltosa, azúcar, lactosa | 5,8 | 1,2 |
| Nestlé crecimiento junior con rico sabor a galleta maría | Desnatada | +12 meses | Azúcar, caramelo | 8,1 | 3,5 |
| Nestlé crecimiento junior con rico sabor a galleta maría | Desnatada | +2 años | Azúcar, caramelo | 6,9 | 2,3 |
| Hacendado energía y crecimiento | Semidesnatada | +3 años | Azúcar, miel | 7 | 2,4 |
| Hacendado crecimiento | Semidesnatada | +12 meses | Sacarosa, dextrinomaltosa, miel | 7,5 | 2,9 |
| Hacendado crecimiento con cereales | Semidesnatada | +12 meses | Sacarosa, dextrinomaltosa, miel | 8,7 | 4,1 |
| Celta sin lactosa semidesnatada crecimiento | Semidesnatada | +3 años | 5,1 | 0,5 | |
| Kaiku sin lactosa peques en crecimiento | Semidesnatada | +3 años | Fructosa | 4,9 | 0,3 |
| Celta leche crecimiento semidesnatada | Semidesnatada | +3 años | 6 | 1,4 | |
| Leche enriquecida energía y crecimiento DIA | Semidesnatada | Sin edad | 5 | 0,4 | |
| Babysmile crecimiento (DIA) | Semidesnatada | +12 meses | Maltodextrina, azúcar | 3,5 | |

| Figura 2. Contenido de azúcares totales en g/100 ml de las leches de crecimiento disponibles en Badajoz (2017) |
|---|
 |

El porcentaje calórico por cada 100 ml de producto que aportaron los azúcares totales osciló entre el 20 y el 48%, la cifra más alta corresponde al producto denominado “Nestlé crecimiento junior bajo contenido en lactosa”, cuyo ingrediente principal es el agua (Tabla 2). El 70% (14/20) de los productos superaron el porcentaje calórico aportado por los azúcares naturalmente presentes en la leche entera de vaca, que asciende al 29%. En cuanto a los azúcares libres, este porcentaje osciló entre el 3 y el 22% del aporte calórico total. De los 14 productos con estimación de azúcares libres, el 50% superaron la cantidad máxima recomendada por la OMS (10% del aporte calórico) para ese nutriente añadido.
| Tabla 2. Porcentaje de energía aportada por los azúcares totales y libres respecto al aporte calórico total de las leches de crecimiento disponibles en Badajoz (2017) | |||||
|---|---|---|---|---|---|
| Presentación comercial | kcal/100 ml* | Azúcares totales (kcal/100 ml) | Porcentaje calórico aportado por los azúcares totales** | Azúcares libres (kcal/100 ml) | Porcentaje calórico aportado por los azúcares libres** |
| Puleva peques 3 crecimiento | 60 | 13,2 | 22 | ||
| Puleva peques 3 crecimiento con cereales | 78 | 16 | 20 | ||
| Puleva peques 3 crecimiento con cereales al cacao | 90 | 27,2 | 30 | 8,8 | 10 |
| Puleva MAX energía + crecimiento | 62 | 26,8 | 43 | 8,4 | 13 |
| Puleva MAX energía + crecimiento con cereales | 77 | 26 | 34 | 7,6 | 10 |
| Puleva MAX energía + crecimiento sin lactosa | 59 | 23,2 | 39 | 4,8 | 8 |
| Nestlé crecimiento junior con todo su sabor original | 70 | 20,8 | 29 | 2,4 | 3 |
| Nestlé crecimiento junior bajo contenido en lactosa | 70 | 34 | 48 | 15,6 | 22 |
| Nestlé crecimiento junior con todo su sabor original | 71 | 20,8 | 29 | 2,4 | 3 |
| Nestlé crecimiento junior con delicioso sabor a cereales | 80 | 23,2 | 29 | 4,8 | 6 |
| Nestlé crecimiento junior con rico sabor a galleta maría(+ 12 meses) | 83 | 32,4 | 39 | 14 | 17 |
| Nestlé crecimiento junior con rico sabor a galleta maría (+2 años) | 77 | 27,6 | 36 | 9,2 | 12 |
| Hacendado energía y crecimiento | 67 | 28 | 42 | 9,6 | 14 |
| Hacendado crecimiento | 65 | 30 | 46 | 11,6 | 18 |
| Hacendado crecimiento con cereales | 82 | 34,8 | 42 | 16,4 | 20 |
| Celta sin lactosa semidesnatada crecimiento | 48 | 20,4 | 42 | 2 | 4 |
| Kaiku sin lactosa peques en crecimiento | 46 | 19,6 | 43 | 1,2 | 3 |
| Celta leche crecimiento semidesnatada | 60 | 24 | 40 | 5,6 | 10 |
| Leche enriquecida energía y crecimiento DIA | 47 | 20 | 42 | 1,6 | 3 |
| Babysmile crecimiento (DIA) | 61 | 14 | 23 | ||

Todas las LC objeto del estudio hacen alegaciones nutricionales, con expresiones del tipo “fuente de” o “enriquecido con”, relativas a la presencia ácidos grasos omega-3 ácido eicosapentaenoico (EPA) y ácido docosahexaenoico (DHA), diversas vitaminas y minerales como calcio, zinc, hierro, yodo, magnesio y fósforo. En el 60% de las presentaciones (12/20) aparecen declaraciones de propiedades saludables que no han sido autorizadas por la Autoridad Europea de Seguridad Alimentaria (EFSA). En concreto, las seis variedades de Puleva® realizan la siguiente declaración no autorizada: “Es fuente de hierro, contribuye al correcto aprendizaje de los niños”, en lugar de: “El hierro contribuye al desarrollo cognitivo normal de los niños”. Las tres variedades de “Puleva Max” presentan otra declaración no aprobada: “Sustituye parte de la grasa saturada por grasa insaturada, como el ácido graso omega-3 DHA presente en la leche materna. Forma parte de las células de la retina”. En las cuatro variedades de “Nestlé crecimiento junior” para mayores de un año de edad, aparece la siguiente declaración no autorizada: “Zinc: ayuda al crecimiento”. Las dos variedades de “Hacendado crecimiento” destinadas a mayores de un año presentan “Composición en grasa, proteína e hidratos de carbono modificada siguiendo las recomendaciones nutricionales para niños”, que no se corresponde con ninguna de las declaraciones nutricionales autorizadas por el Reglamento Europeo.
En las 12 (60%) presentaciones comerciales correspondientes a las marcas Nestlé® y Puleva®, aparece en el frontal del envase la frase: “Nestlé/Puleva colabora con la Asociación Española de Pediatría”, siendo, en el caso de Puleva®, más grandes las siglas de esta asociación (AEP) que la letra del resto de la frase y, en el caso de Nestlé®, más grande y con mayúsculas la palabra “Pediatría”.
Los precios encontrados en este estudio variaron de 0,92 € a 1,67 € por litro, entre dos y tres veces el precio de la leche entera de vaca. Respecto a su colocación dentro de los supermercados, las dirigidas a tres o más años se sitúan en el pasillo de productos lácteos, mientras que las dirigidas a edades comprendidas entre uno y dos años se ubican en el pasillo de alimentación infantil.
DISCUSIÓN
En 2017, las LC disponibles en los supermercados de Badajoz para niños de uno a tres años presentan un alto contenido calórico y de azúcares y bajo de proteínas y grasas, en comparación con la leche entera de vaca. El 85% de las LC analizadas contienen azúcares añadidos en su lista de ingredientes, en contra de las recomendaciones de la OMS, llegando en algunos casos hasta casi doblar el contenido en gramos de azúcares totales de la leche entera de vaca. Los azúcares libres representaron entre el 3 y el 22% del aporte calórico total de las LC. Todas las LC presentaron algún tipo de alegación nutricional o de salud. En 12 variedades (60%) se hizo uso de alegaciones de salud no autorizadas y el mismo número de productos contó con el aval de la AEP.
El mercado de los preparados para niños de corta edad está repartido entre un pequeño número de fabricantes; los radicados en la Unión Europea son los líderes mundiales18. Por tanto, no sorprende que otros estudios que han analizado la composición nutricional de las LC y productos similares para niños pequeños hayan obtenido resultados similares al nuestro en cuanto a composición nutricional y contenido de azúcares19,20. La constatación de la existencia de tres tipos de LC con menor cantidad de azúcares totales que la leche de vaca, pese a constar la presencia de azúcares añadidos en su lista de ingredientes, sugiere que el porcentaje de leche puede ser muy bajo en algunos de estos productos, sin llegar a constituir el componente principal de los mismos, pese a presentarse como LC. De hecho, en uno de los productos con mayor cantidad de azúcares, el ingrediente principal es el agua. Además, el hecho de que tres productos contienen más cantidad de azúcares totales que la leche semidesnatada con que están elaborados, pero sin declarar la presencia de azúcares añadidos en la lista de ingredientes, denota la existencia de errores en el etiquetado u ocultación en la declaración de ingredientes, particularmente en el caso de la variedad “Celta leche crecimiento semidesnatada”; en los otros dos casos, la diferencia con la leche de vaca es tan pequeña que podría deberse a variaciones por azar en la medición. Además de estos posibles errores, al consumidor le resulta imposible conocer la cantidad de azúcares libres presentes en los alimentos mediante la información nutricional del etiquetado, ya que no se separan los azúcares añadidos de los que se encuentran naturalmente en el alimento, teniendo que ir a la lista de ingredientes para ver si dichos alimentos llevan o no azúcares añadidos. Puesto que son los azúcares añadidos los que se han asociado con la obesidad, la caries y la presencia de factores de riesgo cardiovascular, resulta imprescindible establecer medidas relativas al contenido nutricional de estos productos y a la regulación de su presentación y publicidad. Sería conveniente implantar un etiquetado interpretativo más comprensible para el consumidor, y distinguir la cantidad de azúcares presentes de forma natural en el alimento de la cantidad de azúcares añadidos o liberados durante el proceso tecnológico.
Todas las LC objeto de este estudio presentan alegaciones nutricionales por su contenido en vitaminas y minerales, cuyas necesidades pueden ser perfectamente cubiertas mediante una dieta equilibrada sin necesidad de enriquecimiento de sus productos, que resulta por tanto innecesario y en algunos casos, como el calcio y la vitamina D, puede llegar a afectar negativamente a la salud de los niños si se consumen en exceso21. Además, un estudio reciente ha mostrado que los niños que consumen leche entera de vaca tienen menor ganancia de peso que aquellos que la consumen desnatada, logrando además mayores niveles de vitamina D22. Por el contrario, el consumo de LC, muy frecuente entre los 12 y los 18 meses, se ha asociado con un aumento del riesgo de desarrollar obesidad en niños23.
La aparición de declaraciones de propiedades saludables no autorizadas por la EFSA constituye un ejemplo de publicidad engañosa. Este tipo de fraude tiene gran relevancia ya que la principal razón alegada para consultar el etiquetado por la mayoría de los consumidores fue elegir productos más saludables24. Además, esto les permite aumentar el precio del producto, hasta triplicar en algunos casos el de la leche entera de vaca, aprovechando que el consumidor está dispuesto a pagar más por productos con alegaciones que les aportan un halo saludable25. El aval de la AEP, aunque legalmente permitido, también constituye un tipo de publicidad engañosa y cuestionable desde el punto de vista de la ética profesional, cuando se otorga, como en el caso que nos ocupa, a productos que no cumplen las recomendaciones de la OMS y de los expertos7,8,13,14. Los consumidores, totalmente indefensos ante la presencia de alegaciones de salud sin fundamento científico que las respalde y el uso engañoso de alegaciones nutricionales y avales de asociaciones de profesionales de la salud, son inducidos a error por el halo saludable que rodea a las LC, pese a ser más caras y menos recomendables para sus hijos que la leche entera de vaca.
La EFSA elaboró en 2013 un informe técnico, a petición de la Comisión Europea, donde concluyó que las LC constituyen un modo de aumentar el aporte de ácidos grasos poliinsaturados omega-3, hierro y vitamina D en lactantes y niños de corta edad, pero indicando que existen alternativas eficaces para aumentar el aporte de esos nutrientes26,27. Finalmente, en su dictamen de 26 de junio de 2014, la EFSA señaló que los niños de corta edad pueden seguir tomando los preparados de inicio y de continuación consumidos durante el primer año de vida, por lo cual no consideró necesario proponer una composición específica de las LC27. Sin embargo, un informe externo solicitado por la EFSA ha constatado la presencia extendida de LC en el mercado de todos los países de la Unión Europea, con un consumo creciente en prácticamente todos ellos. Los países con mayor consumo y número de productos fueron Francia, España, Italia y Alemania18. El alto contenido de azúcares de estos productos, en contra de las recomendaciones de la OMS y de los expertos, pone de manifiesto la necesidad de establecer una regulación específica de su composición28, al objeto de proteger a los niños durante su primera infancia, un periodo de especial vulnerabilidad. Además, el Reglamento de Declaraciones de Propiedades Saludables establecía en su preámbulo el futuro desarrollo de perfiles nutricionales, para que solo los alimentos y bebidas con un perfil nutricional saludable puedan llevar declaraciones de salud. En el caso de LC, esto implicaría la prohibición del uso de alegaciones y avales científicos o de grupos profesionales de la salud en la práctica totalidad de las variedades disponibles actualmente en el mercado, por la presencia de azúcares añadidos.
El tamaño muestral pequeño es una posible limitación de este estudio, al haber sido obtenido en los supermercados de la ciudad de Badajoz. Sin embargo, las cadenas de supermercados visitadas son la mayoría de las presentes a nivel nacional y los productos ofertados son producidos por un reducido número de multinacionales alimentarias, por lo que no varía mucho su oferta entre supermercados, y no es previsible que el tamaño y composición de la muestra hubiese sido muy diferente de haber incluido otras ciudades. Otra limitación consiste en la ausencia de información sobre contenido en azúcares añadidos en la tabla de composición nutricional del etiquetado, que nos ha obligado a estimar la cantidad de azúcares libres presentes en las LC. Puesto que el dato se ha obtenido de la diferencia en azúcares totales con la leche de vaca y el porcentaje de leche que contiene cada producto es desconocido, la cantidad de azúcares libres podría estar en algunos casos infraestimada.
En conclusión, consideramos necesario regular las LC, cuyo consumo se está extendiendo entre los niños pequeños, por su alto contenido en azúcares añadidos, incompatible con las recomendaciones de la OMS y los expertos en nutrición. La regulación debería contemplar la composición nutricional de los productos, un etiquetado más informativo, de tipo interpretativo, y la prohibición del uso de alegaciones nutricionales o de salud y de avales científicos en productos dirigidos a niños pequeños que no se ajusten a las recomendaciones nutricionales vigentes y, de forma particular, a los que contengan azúcares añadidos.
NOTA DE LOS AUTORES
Este artículo presenta resultados o investigación independientes. Las opiniones expresadas son las de los autores y no representan necesariamente la posición oficial del Instituto de Salud Carlos III.
CONFLICTO DE INTERESES
Los autores declaran no presentar conflictos de intereses en relación con la preparación y publicación de este artículo.
ABREVIATURAS
AEP: Asociación Española de Pediatría; BEDCA: Base de Datos Española de Composición de Alimentos; DHA: ácido docosahexaenoico; EFSA: Autoridad Europea de Seguridad Alimentaria; EPA: ácido eicosapentaenoico; LC: leches de crecimiento; OMS: Organización Mundial de la Salud.
BIBLIOGRAFÍA
- Sánchez Cruz JJ, Jiménez Moleón JJ, Fernández Quesada F, Sánchez MJ. Prevalence of child and youth obesity in Spain in 2012. Rev Esp Cardiol (Engl Ed). 2013;66:371-6.
- Ramiro González MD, Sanz Barbero B, Royo Bordonada MA. Childhood excess weight in Spain from 2006 to 2012. Determinants and parental misperception. Rev Esp Cardiol. 2017;70:656-63.
- Monasta L, Batty GD, Cattaneo A, Lutje V, Ronfani L, Van Lenthe FJ, et al. Early-life determinants of overweight and obesity: a review of systematic reviews. Obes Rev. 2010;11:695-708.
- Vos M, Kaar J, Welsh J, Van Horn L, Feig D, Anderson C, et al. Added sugars and cardiovascular disease risk in children: a scientific statement from the American Heart Association. Circulation. 2016;135:e1017-e1034.
- Drewnowski A, Mennella JA, Johnson SL, Bellisle F. Sweetness and food preference. J Nutr. 2012;142:1142S-1148S.
- Tryon M, Stanhope K, Epel E, Mason A, Brown R, Medici V, et al. Excessive sugar consumption may be a difficult habit to break: a view from the brain and body. J Clin Endocrinol Metabol. 2015;100:2239-47.
- Informe del Consejo Ejecutivo sobre su 106.ª y 107.ª reuniones de la OMS. A54/2. 30 de marzo de 2001. En: Organización Mundial de la Salud [en línea] [consultado el 06/11/2018]. Disponible en http://apps.who.int/gb/archive/pdf_files/WHA54/sa542.pdf
- Conclusiones del Consejo para contribuir a detener el aumento de sobrepeso y la obesidad infantil 2017/C205:46-52. En: Diario Oficial de la Unión Europea [en línea] [consultado el 06/11/2018]. Disponible en http://eur-lex.europa.eu/legal-content/ES/TXT/PDF/?uri=CELEX:52017XG0629(01)&from=ES
- Resolución WHA63.14 sobre la promoción de alimentos y bebidas no alcohólicas dirigida a los niños, aprobado por la 63.ª Asamblea Mundial de la Salud. En: Organización Mundial de la Salud [en línea] [consultado el 06/11/2018]. Disponible en http://apps.who.int/gb/ebwha/pdf_files/WHA63/A63_R14-sp.pdf
- Moynihan P, Kelly S. Effect on caries of restricting sugars intake. J Dental Res. 2013;93:8-18.
- Te Morenga L, Mallard S, Mann J. Dietary sugars and body weight: systematic review and meta-analyses of randomised controlled trials and cohort studies. BMJ. 2012;346:e7492-e7492.
- Malik VS, Pan A, Willett WC, Hu FB. Sugar-sweetened beverages and weight gain in children and adults: a systematic review and meta-analysis. Am J Clin Nutr. 2013;98:1084-102.
- Galiano Segovia MJ, Moreno Villares JM. La leche de vaca en la alimentación del niño: ¿necesaria o causa de problemas? Pediatr Integral. 2013;17:371-6.
- Dalmau J. Nutrición en la infancia y en la adolescencia. En: Carbajal A, Martínez C (coords.). Manual práctico de nutrición y salud. Madrid: Exlibris Ediciones; 2012. p. 207-21.
- Informe de la Comisión al Parlamento Europeo y al Consejo sobre los preparados para niños de corta edad. Bruselas: 31.3.2016. COM (2016) 169 final. En: Comisión Europea [en línea] [consultado el 06/11/2018]. Disponible en https://ec.europa.eu/transparency/regdoc/rep/1/2016/ES/1-2016-169-ES-F1-1.PDF
- Sugars intake for adults and children. En: Organización Mundial de la Salud [en línea] [consultado el 06/11/2018]. Disponible en who.int/nutrition/publications/guidelines/sugars_intake/en/
- Basulto J, Ojuelos FJ, Baladia E, Manera M. Azúcares en alimentos infantiles. La normativa española y europea, ¿a quién protege? Rev Pediatr Aten Primaria. 2016;18:e47-e53.
- AINIA Centro Tecnológico. Report of data collection with respect to the availability and nutritional composition of different types of milk-based drinks and similar products for young children with the denomination of “growing up milks” or “toddlers milk” or with similar terminology currently on the market in EU member States. EFSA supporting publication 2013: EN-505. En: EFSA Online Library [en línea] [consultado el 06/11/2018]. Disponible en http://onlinelibrary.wiley.com/doi/10.2903/sp.efsa.2013.EN-505/epdf
- La alimentación industrializada del lactante y el niño pequeño. El nuevo meganegocio. El poder de consumidor. En: Ministerio de Salud. Gobierno de Costa Rica [en línea] [consultado el 06/11/2018]. Disponible en ministeriodesalud.go.cr/gestores_en_salud/lactancia/articulos/CNLM_alimentacion_industrializada_lactante_nino_pequeno.pdf
- Jardí C, Aranda N, Bedmar C, Arija V. Composición nutricional de las leches infantiles. Nivel de cumplimiento en su fabricación y adecuación a las necesidades nutricionales. An Pediatr (Barc). 2015;83:417-29.
- Molina H, Mena P, Vial P, Fernández ME, Alcázar ML, Muzzo S. Intoxicación por vitamina D en el lactante. Rev Chil Pediatr. 1984;55:270-3.
- Morency M, Birken C, Lebovic G, Chen Y, L’Abbé M, Lee G, et al. Association between noncow milk beverage consumption and childhood height. Am J Clin Nutr. 2017;106:597-602.
- Wiberger M, Eiben G, Lissner L, Mehlig K, Papoutsou S, Hunsberger M. Children consuming milk cereal drink are at increased risk for overweight: the IDEFICS Sweden study, on behalf of the IDEFICS Consortium. Scan J Public Health. 2014;42:518-24.
- Prieto Castillo L, Royo Bordonada MA, Moya Geromini A. Information search behaviour, understanding and use of nutrition labeling by residents of Madrid, Spain. Public Health. 2015;129:226-36.
- De Magistris T, López Galán B. Consumers’ willingness to pay for nutritional claims fighting the obesity epidemic: the case of reduced-fat and low salt cheese in Spain. Public Health. 2016;135:83-90.
- Panel on Dietetic Products, Nutrición and Allergies (NDA). Scientific opinion on nutrient requirements and dietary intakes of infant and young children in the European Union. EFSA J. 2013;11:3408.
- EFSA Panel on Dietetic Products, Nutrición and Allergies (NDA). Scientific opinion on the essential composition of infant and follow-on formulae. EFSA J. 2014;12:3760.
- Palou Óliver, A, Palou March M. La evidencia científica la información al consumidor: las declaraciones nutricionales y de propiedades saludables (health claims) en los alimentos. Rev Esp Comun Salud. 2016;S1:31-42.




