Vol. 20 - Num. 79
Originales
Deshidratación hipernatrémica grave neonatal por fallo en la instauración de la lactancia materna: estudio de incidencia y factores asociados
David López Martína, M.ª Mar Alonso Montejob, José Miguel Ramos Fernándeza, Ana M.ª Cordón Martíneza, Tomás Sánchez Tamayoc, Antonio Luis Urda Cardonaa
aUGC de Pediatría. Hospital Materno-Infantil de Málaga. Hospital Regional Universitario de Málaga. Málaga. España.
bMIR-Pediatría. Hospital Materno Infantil de Málaga. Málaga. España.
cUGC de Neonatología. Hospital Materno-Infantil de Málaga. Hospital Regional Universitario de Málaga. Málaga. España.
Correspondencia: D López. Correo electrónico: lopez.martin.david@hotmail.com
Cómo citar este artículo: López Martín D, Alonso Montejo MM, Ramos Fernández JM, Cordón Martínez AM, Sánchez Tamayo T, Urda Cardona AL. Deshidratación hipernatrémica grave neonatal por fallo en la instauración de la lactancia materna: estudio de incidencia y factores asociados . Rev Pediatr Aten Primaria. 2018;20:229-35.
Publicado en Internet: 10-09-2018 - Número de visitas: 43787
Resumen
Introducción: la deshidratación hipernatrémica neonatal asociada al fallo en la instauración de la lactancia materna puede provocar importantes complicaciones e incluso la muerte. Existen pocos datos acerca de su magnitud en nuestro medio. Objetivos: conocer la incidencia y describir las características clínico-epidemiológicas asociadas.
Material y métodos: estudio observacional retrospectivo en recién nacidos con diagnóstico de deshidratación hipernatrémica con Na>150 mEq/l que necesitaron ingreso hospitalario entre 2011 y 2017 en nuestra área sanitaria. Se excluyeron casos con patología de base o infección concomitante. Se recogieron datos demográficos, clínicos, analíticos, terapéuticos y evolutivos mediante revisión de historias clínicas y se realizó análisis estadístico posterior.
Resultados: la población susceptible estimada fue de 41 084 recién nacidos. Cumplieron criterios 20 casos. Alimentación con lactancia materna exclusiva 19/20, primer hijo en 14/20 familias, con mediana de edad materna 34 años (rango intercuartílico: 31-37). Los motivos de consulta más frecuentes fueron pérdida de peso, ictericia e irritabilidad. La mediana de edad al ingreso fue de 5,5 días (rango intercuartílico: 3,3-9,8), con porcentaje de peso perdido 14,4% (rango intercuartílico: 10-17) y natremia mediana 157,25 mEq/L (rango intercuartílico: 152-157,8). La estancia media fue de 6,5 días (rango intercuartílico: 4-8,75), 2/20 precisaron cuidados intensivos. No registramos fallecimientos o complicaciones graves.
Conclusiones: la incidencia global de la deshidratación hipernatrémica fue de 0,5 por cada 1000 recién nacidos/año en la población estudiada. Se relacionó con lactancia materna exclusiva, primiparidad y mayor edad materna. Con estos resultados, parece una medida oportuna informar sobre signos de alarma al alta de maternidad, así como realizar la primera revisión del bebé de forma precoz.
Palabras clave
● Deshidratación ● Hipernatremia ● Lactancia materna ● Recién nacidoINTRODUCCIÓN
La deshidratación hipernatrémica en neonatos (DHN) alimentados con lactancia materna (LM) exclusiva es una entidad que comienza a describirse a partir de 1990. La DHN es un proceso potencialmente grave1,3 que puede provocar secuelas neurológicas importantes e incluso la muerte4. La incidencia real de la DHN en la LM exclusiva es poco conocida en nuestro medio, aunque por las consecuencias se trata de un verdadero problema de salud pública; se ha estimado en 1,7-5/1000 recién nacidos (RN) vivos2,5.
La DHN se define por niveles de sodio en sangre ≥150 mmol/l, pudiendo estar asociado a pérdida de peso superior a la fisiológica (10%)4,6. Se presenta hacia los ocho días de vida, con un rango encontrado en los diferentes estudios de 2 a 14 días.
Es una entidad infradiagnosticada, por presentar síntomas inespecíficos como la letargia, aunque otros pacientes se muestran irritables. Además, pueden presentar signos de deshidratación (como mucosas secas, fontanela deprimida, pobre turgencia de la piel), aspecto de desnutrición, ictericia, fiebre, oligoanuria o alteraciones neurológicas.
Se han descrito como factores de riesgo la administración inapropiada de la alimentación a los neonatos (fallo en la técnica de LM, déficit succión o hipogalactia), la prematuridad, la edad gestacional pequeña o bajo peso al nacer, madre primípara, cesárea o que las madres no identifiquen los signos de baja ingesta de manera temprana4,7-10. La disminución de la orina al día (menos de seis cambios de pañal), menos de cuatro deposiciones al día a partir del cuarto día de vida, la presencia de cristales de urato en la orina después del tercer día, la hiperbilirrubinemia y una pérdida mayor del 10% son factores de mal pronóstico11.
Algunos autores han encontrado relación entre la DHN y los elevados niveles de sodio en la leche materna en madres con dificultades en la LM, no produciéndose el descenso fisiológico de este ión2,12.
El objetivo del estudio fue determinar la incidencia de ingresos por DHN grave y describir los factores de riesgo asociados para poder proponer medidas preventivas.
MATERIAL Y MÉTODOS
Estudio descriptivo retrospectivo de pacientes menores de 30 días ingresados en un hospital de referencia entre enero del 2011 y diciembre del 2016 con diagnóstico de deshidratación hipernatrémica (natremia ≥150 mmol/l). Se incluyeron a recién nacidos sanos a término (37-42 semanas de edad gestacional) menores de 30 días de vida, dados de alta a su domicilio tras el nacimiento y que precisaron ingreso, pertenecientes a nuestra área de salud.
Se excluyeron pacientes con antecedentes de prematuridad, patología infecciosa concomitante, patología de base y exitus sin causa conocida en el momento de realizar el estudio. Los datos se obtuvieron mediante revisión de las historias clínicas de los RN ingresados en nuestro hospital.
La variable principal fue el nivel sérico de sodio determinado en una muestra venosa. Otras variables consideradas fueron edad materna, número de gestación, tipo de parto, género, edad gestacional, edad al ingreso, peso al nacimiento, al ingreso y al alta, temperatura al ingreso, tipo de alimentación recibida, presencia de datos clínicos de deshidratación, creatinina, nitrógeno ureico en sangre (BUN) y urea del ingreso, niveles de glucosa sérica al ingreso, derivación a nuestro centro, valoración previa por profesional de la salud, los días de estancia intrahospitalaria, ingreso en unidad de cuidados intensivos pediátricos (UCIP), necesidad de otros estudios complementarios, presencia de complicaciones, comorbilidades y mortalidad.
Además, se clasificó el motivo de consulta de los padres al centro sanitario según síntomas: ictericia, irritabilidad, somnolencia, ausencia de deposiciones en 24 horas, fiebre, rechazo de la alimentación, infarto úrico, número de cambios de pañal al día y pérdida de peso. El estudio fue posible por la estandarización informatizada de nuestra historia clínica.
Se contrastaron las principales variables epidemiológicas con los datos previamente publicados.
Se realizó un estudio estadístico con SPSS® v.22 considerando una p < 0,05 para la significación estadística. Los resultados de las variables cualitativas fueron expresados como frecuencias con intervalos de confianza; y los de las cuantitativas mediante mediana y rango intercuartílico.
Se estimó la población susceptible según la adscripción poblacional de nuestra área de salud para la obtención del cálculo de la incidencia. Se comparó la diferencia de esta durante estaciones cálidas (primavera y verano) y estaciones frías (otoño e invierno) mediante t de Student.
El trabajo se realizó de acuerdo con los criterios del comité de ética de nuestro centro. Respecto a las normas de confidencialidad, los datos clínicos se obtuvieron de forma disociada.
RESULTADOS
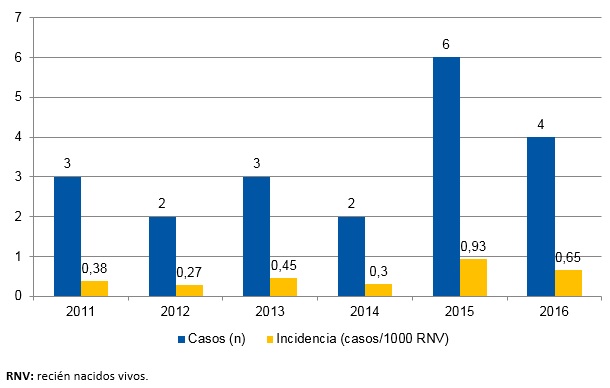
La población susceptible estimada fue de unos 41 084 recién nacidos durante el periodo de estudio, en nuestro caso correspondiente a los nacidos en las maternidades de un hospital de segundo y tercer nivel, ambos centros hospitalarios del sistema sanitario público del área de influencia de nuestra muestra. Cumplieron criterios del estudio 20 casos de entre los 22 neonatos dados de alta con el diagnóstico codificado de deshidratación hipernatrémica; dos sujetos fueron excluidos por presentar patología desencadenante (hipercoagulabilidad, infección del tracto urinario [ITU]). La incidencia promedio durante el periodo de estudio fue de 0,5 por cada 1000 RN/año, desglosada por años en la Fig. 1, siendo máxima en el año 2015 (0,93 por 1000 RN/año) y mínima en el año 2012 (0,27 por 1000 RN/año).
Los datos demográficos y características neonatales y maternas quedan recogidos en la Tabla 1. Entre el total de la muestra, había 11 niñas (55%) y 9 niños (45%). Ocho casos (40%) tuvieron lugar durante primavera-verano y 12 (60%) en otoño-invierno, sin ser estas diferencias estadísticamente significativas (p = 0,371). La edad gestacional media de nuestros pacientes fue de 39,1 semanas (desviación estándar [DE] ±1,11) con peso medio al nacimiento de 3272 g (±373,3 DE). Nacieron por cesárea dos de los casos (1%), indicadas por registro fetal no tranquilizador y por no progresión respectivamente. Los 20 recién nacidos (100%) presentaron una puntuación de entre 9-10 en el test de Apgar a los 5 y 10 minutos de vida. El alta de la planta de maternidad tras el nacimiento se produjo entre el 2.º y 3.º día de vida en 18 de los casos (90%); en ninguno de los casos fue precoz (tras 24 horas). Entre los pacientes, 14 eran el primer hijo (el 70% fueron primíparas) y el valor mediano de la edad materna en la muestra fue de 34 años (rango intercuartílico [RIQ]: 31-37). La gran mayoría, 19 (95%) fueron alimentados mediante LM exclusiva –una de estas madres presentaba mastitis unilateral en el momento del ingreso del bebé–, solo en uno de los casos la LM era suplementada desde el nacimiento con fórmula adaptada por decisión familiar.
Cinco de los bebés (25%) acudían derivados tras valoración previa, en tres de los casos por su pediatra, en uno de los casos por matrona y en otro de los casos por un equipo de emergencias. En los casos restantes la familia consultó en urgencias de nuestro centro por criterio propio. El motivo de consulta se enumera en la Tabla 1: los más frecuentes fueron la presencia de ictericia –en cuatro casos (20%) en rango de fototerapia–, e irritabilidad con llanto no consolable, seguidos por somnolencia, ausencia de deposiciones en 24 horas, fiebre y pérdida de peso excesiva o no recuperación del peso al nacimiento. Otros motivos de consulta menos frecuentes fueron rechazo de la alimentación y presencia de infarto úrico. La mediana de edad al ingreso fue de 5,5 días (RIQ: 3,3-9,8), y la mediana de cambios de pañal fue de tres al día (RIQ: 2-4). La mediana del porcentaje de pérdida de peso con respecto al nacimiento del 14,4% (RIQ: 10-17). En la exploración a su llegada, 11 de los pacientes (55%) presentaban una succión ineficaz, lenta o perezosa.
| Tabla 1. Características neonatales y maternas (n = 20) en la deshidratación hipernatrémica neonatal por lactancia materna | |||
|---|---|---|---|
| Variable (unidad de medida) | Mediana | Rango intercuartílico | Rango |
| Edad al ingreso (días) | 5,5 | 3,3-9,8 | 2-15 |
| N.º pañales orina/día | 3 | 2-4 | 0-6 |
| N.º pañales deposiciones/día | 1 | 0-1 | 0-4 |
| Pérdida de peso (%) | 14,4 | 10-17 |
5,9-30 |
| Niveles de sodio sérico (mEq/l) | 155 | 152-157,8 | 150-178 |
| Edad materna (años) | 34< | 31-37 | 19-47 |
| Variable (unidad de medida) | Media | Desviación estándar | |
| Peso al nacimiento, media (gramos) | 3272,3 | ±373,3 | |
| Edad gestacional media (semanas) | 39,1 | ±1,11 | |
| Variable | N.º de casos | ||
| Sexo femenino del paciente | 11 | ||
| Lactancia materna exclusiva | 19 | ||
| Mala succión | 11 | ||
| Parto eutócico | 18 | ||
| Madre primípara | 14 | ||
| Mastitis materna | 1 | ||
| Ingreso durante la estación fría |
12 |
||
| Motivo de consulta al ingreso (n = 20) | |||
| Irritabilidad | 9 | ||
| Ictericia | 9 | ||
| Somnolencia | 8 | ||
| Ausencia de deposiciones en 24 horas | 8 | ||
| Fiebre | 7 | ||
| Pérdida de peso | 6 | ||
| Rechazo alimentación | 4 | ||
| Infarto úrico | 4 | ||

La mediana para la cifra de sodio en sangre fue de 157,25 mEq/l (RIQ: 152-157,8). Presentaron acidosis metabólica leve 12 de ellos (60%), con cifras medianas de pH en la muestra de 7,34 (RIQ: 7,29-7,35) e hipoglucemia en cinco (25%) con cifras medianas de glucemia en la muestra de 65 mg/dl (RIQ: 53-82). La cifra mediana de creatinina en nuestra serie fue de 0,62 mg/dl (RIQ: 0,53-0,73). Se realizó ecografía craneal en 12 (60%) de los neonatos –bien por somnolencia excesiva (siete de ellos) o bien por irritabilidad (cinco)–, presentando alteraciones en la prueba tan solo uno de ellos –leve hiperecogenicidad en núcleos de la base de forma bilateral, con posible edema–; aunque no se efectuó prueba de imagen de control, la evolución neurológica de este paciente fue satisfactoria. Los datos recogidos en otras pruebas complementarias se reflejan en la Tabla 2.
| Tabla 2. Otras alteraciones en las pruebas complementarias (n = 20) en la deshidratación hipernatrémica neonatal por lactancia materna | |||
|---|---|---|---|
| Variable | N.º de casos | ||
| Acidosis metabólica leve | 12 | ||
| Insuficiencia renal prerrenal | 5 | ||
| Glucemia <60 mg/dl | 5 | ||
| Alteraciones en ecografía craneal | 1 | ||

La estancia media fue de 6,5 días (RIQ: 4-8,75), con una mediana de peso al alta de 3175 g (RIQ: 2800-3370). Dos de ellos requirieron ingreso en cuidados intensivos durante 48 horas, por natremia de 178 mEq/l y por clínica neurológica con apneas centrales respectivamente. Necesitaron fluidoterapia intravenosa 16 de ellos (80%) con un tiempo promedio para la corrección de la hipernatremia de 48 h, sin precisar ninguno técnicas de depuración extrarrenal, y todos ellos salvo uno recibieron fórmula adaptada durante el ingreso, aunque solamente se evidenció hipogalactia materna en 13 casos (65%). No tuvimos ningún fallecimiento en nuestra serie.
DISCUSIÓN
La cifra global de la DHN en nuestra muestra (0,5 por cada 1000 RN al año) fue muy cercana a la descrita en la literatura médica. Como datos de referencia en nuestro país, la incidencia de hipernatremia por mil recién nacidos vivos en un estudio en Asturias fue de 0,47/1000 entre 2002 y 2008 y de 0,81/1000 entre 2009 y 201513, aunque este trabajo incluye recién nacidos desde maternidad, más fáciles de detectar. En el hospital de Gandía (Valencia) fue de 1,4 en el año 2002 con un pico hasta 10,3 en el año 200014. En contraste con la mejora en otros aspectos asistenciales, no encontramos una tendencia al descenso de casos en nuestra serie a lo largo del periodo recogido, ni tampoco en relación con lo previamente publicado, lo que nos plantea la necesidad de estrategias de abordaje de un problema aún no resuelto.
En nuestra serie todos los casos se produjeron en los primeros 15 días de vida, con una edad mediana al ingreso algo mayor que la reflejada en otras series, probablemente debido a que nuestro trabajo se centra en recién nacidos tras el alta.
Los motivos de consulta predominantes en nuestra casuística fueron por igual irritabilidad e ictericia, presentación acorde con la literatura médica, donde también se destacan la hipertonía e hiperreflexia, alteraciones del estado de conciencia o convulsiones que no observamos entre nuestros pacientes14,15. Al igual que en casuísticas previas, también destacamos la importancia de la pérdida de peso desde el nacimiento, así como la frecuencia en la aparición de cristales de urato en la orina14,16-18.
En consonancia con otras series, no encontramos antecedentes obstétricos y perinatales de patología entre nuestros recién nacidos19. Al contrario que en otras publicaciones, no encontramos relación con la realización de cesárea, ya que por lo general no suele suponer en nuestro medio un retraso del inicio de la LM mayor de 12 horas20. La mayoría de las madres fueron primíparas, con una mediana de edad de 34 años, coincidiendo con estudios previos en el que la edad mediana de las mismas fue de 30 años17 y 34 años13.
Entre las comorbilidades metabólicas asociadas detectamos hallazgos bastante similares a otras publicaciones en nuestro medio, con acidosis leve en la mitad de los casos, aunque sin la aparición de trastornos durante la corrección descritos en otras series21,22. Nuestra serie registró un escaso número de convulsiones o alteraciones intracraneales, posiblemente debido a cifras medianas de natremia de 155 mEq/l, inferiores a las series en las que sí se describen13,14.
En nuestro programa de atención al niño sano la primera visita se realiza a los 15 días de vida, que es a nuestro entender muy tardía para detectar problemas derivados de la LM en recién nacidos en riesgo, como reflejan nuestros datos. La planificación de una primera visita al final de la primera semana de vida podría servir para evitar o al menos disminuir los casos de DHN por LM, así como para apoyar la misma evitando casos de renuncia a la LM por técnica ineficaz o claudicación materna13. También podría establecerse una visita el mismo día que se realice el test del talón para pesar al niño, al igual que establecer programas de educación materna en las maternidades13,18. La formación del personal sanitario mediante la creación de documentos de consenso y talleres de apoyo a las madres fueron medidas exitosas que redujeron la incidencia en otras unidades nacionales13,19.
Una de las limitaciones de nuestro estudio, debido a su diseño retrospectivo, es no disponer de datos sobre el nivel educativo de las familias. Un futuro estudio prospectivo, incluyendo el impacto de nuestras propuestas de mejora, podría constituir el punto de partida para disminuir este problema de salud.
Nuestro trabajo es el primero que recoge en nuestro país la incidencia de DHN en recién nacidos tras el alta, destacando la primiparidad como factor de riesgo materno. En la mayoría de los casos los motivos de consulta se relacionaban con problemas asociados a la LM y con signos identificables en una exploración básica. Consideramos que la LM es la mejor manera de alimentar al recién nacido, pero una adecuada monitorización, con una primera visita de niño sano de forma precoz, a los siete días de vida, podría reducir la incidencia del fracaso de la LM y de la DHN.
CONFLICTO DE INTERESES
Los autores declaran no presentar conflictos de intereses en relación con la preparación y publicación de este artículo.
ABREVIATURAS
BUN: nitrógeno ureico en sangre · DE: desviación estándar · DHN: deshidratación hipernatrémica · ITU: infección del tracto urinario · LM: lactancia materna · RIQ: rango intercuartílico · RN: recién nacidos · UCIP: unidad de cuidados intensivos pediátricos.
BIBLIOGRAFÍA
- Neifert MR. Prevention of breastfeeding tragedies. Pediatr Clin North Am. 2001;48:273-97.
- Ivan Amerongen RH, Moretta AC, Gaeta TJ. Severe hypernatremic dehydration and death in a breast-fed infant. Pediatr Emerg Care. 2001;17:175-80.
- Gebara BM, Everett KO. Dural sinus thrombosis complicating hypernatremic dehydration in a breastfed neonate. Clin Pediatr (Phila). 2001;40:45-8.
- Moritz ML, Manole MD, Bogen DL, Ayus JC. Breastfeeding-associated hypernatremia: are we missing the diagnosis? Pediatrics. 2005;116:343-7.
- Segovia RE. Hiponatremia e hipernatremia. En: Facultad de Medicina, Universidad de Chile [en línea] [consultado el 03/09/2018]. Disponible en basesmedicina.cl/nefrologia/12_3_hiponatremia/inicio.htm
- Oddie S, Richmond S, Coulthard M. Hypernatraemic dehydration and breast feeding: a population study. Arch Dis Child. 2001;85:318-20.
- Laing A, Wong C. Hypernatraemia in the first few days: is the incidence rising? Arch Dis Child Fetal Neonatal. 2002;87:F158-F162.
- Moritz ML, Ayus JC. Disorders of water metabolism in children: hyponatremia and hypernatremia. Pediatr Rev. 2002;23:371-80.
- Asturizaga MA, Mazzi GE. Hipernatremia neonatal: factores de riesgo. Rev Chil Pediatr. 2011;82:150-1.
- Erdeve O, Atasay B, Arsan S. Hypernatraemic dehydration in breastfed infants: is caesarean section a risk? Ann Trop Paediatr. 2005;25:147-8.
- Solá A, León HAP. Balance hidroelectrolítico. En: Docstoc [en línea]. Disponible en docstoc.com/docs/153840669/Balance-Hidroelectrol%C3%ADtico-Neonatal
- Juliao J. Deshidratación hipernatrémica e hiperbilirrubinemia indirecta en el recién nacido a término. Rev Medica Sanitas. 2009;12:34-42.
- González García LG, Carrera García L, Arias Llorente RP, Romero MC, Suárez Rodríguez M, Fernández AI, et al. Deshidratación hipernatrémica asociada a la alimentación con lactancia materna en el periodo neonatal. Acta Pediatr Esp. 2016;74:261-5.
- Peñalver Giner O, Gisbert Mestre J, Casero Soriano J, Bernal Ferrer A, Oltra Benavent M, Tomás Vila M. Deshidratación hipernatrémica asociada a lactancia materna. An Pediatr (Barc). 2004;61:340-3.
- Jonguitud-Aguilar A, Calvillo-Robles S, Ruiz-Martínez E, Olvera-López G. Protocolo de manejo en deshidratación hipernatrémica neonatal. Perinatol Reprod Hum. 2015;29:65-9.
- Fernando C, Álvarez H, Fernando J, Robledo G, López AV. Curso clínico de la deshidratación hepernatrémica en recién nacidos. Arch Investig Matern Infant. 2014;6:52-60.
- Berger-Larrañaga M, Bustamante-Abuid C, Díaz-Vergara S, Tresierra-Cabrera J, Mayta-Tristán P, Segura ER. Trastornos de la lactancia materna y otros factores asociados a la pérdida de peso neonatal excesiva en un hospital de la Seguridad Social en Lima, Perú. Nutr Hosp. 2015;32:2062-70.
- Tofé Valera I, Párraga Aquiles MJ, Ruiz González MD, Huertas Muñoz MD, Guzmán Cabañas JM, Zapatero Martínez M. Deshidratación hipernatrémica en un recién nacido con lactancia materna exclusiva. Vox Peadiatr. 2005;1:39-41.
- Vázquez I, Ferrández MJ, Mendoza MR, Quiles JL. Lactancia materna y deshidratación neonatal. ¿Se puede disminuir el número de casos? Acta Pediatr Esp. 2015;73:e247-e251.
- Lavagno C, Camozzi P, Renzi S, Lava SA, Simonetti GD, Bianchetti MG, et al. Breastfeeding-associated hypernatremia: a systematic review of the literature. J Hum Lact. 2016;32:67-74.
- Albalate Ramon M, Alcazar Arroyo R, de Sequera Ortiz P. Alteraciones del agua y del sodio. En: Nefrología al Día [en línea] [consultado el 03/09/2018]. Disponible en revistanefrologia.com/es-monografias-nefrologia-dia-articulo-alteraciones-del-sodio-del-agua-19
- Jaramillo I, López G, Hernández H. Hypernatremic dehydration and death in an infant. Pediatr Emerg Care. 2003;19:62-3.