Vol. 19 - Num. 74
Originales
Seguimiento de los resultados de Microbiología y del tratamiento antibiótico empírico prescrito en un Servicio de Urgencias
Estíbaliz Onís Gonzáleza, Lucila Madariaga Torresb, I Varona Péreza, María Gil Péreza, Esperanza Moya Calderóna, Pilar Embid Pardoa
aPediatra. Clínica IMQ Zorrotzaurre. Bilbao. España.
bDepartamento de Inmunología, Microbiología y Parasitología. Facultad de Medicina y Enfermería. Universidad de País Vasco. Bilbao. España.
Cómo citar este artículo: Onís González E, Madariaga Torres L, Varona Pérez I, Gil Pérez M, Moya Calderón E, Embid Pardo P. Seguimiento de los resultados de Microbiología y del tratamiento antibiótico empírico prescrito en un Servicio de Urgencias. Rev Pediatr Aten Primaria. 2017;19:113-8.
Publicado en Internet: 06-06-2017 - Número de visitas: 18227
Resumen
Introducción: la gestión deficiente de los resultados del Laboratorio de Microbiología tiene un efecto negativo en la seguridad del paciente y en el manejo adecuado de los antibióticos. El objetivo de este estudio es analizar el seguimiento de los resultados microbiológicos en un Servicio de Urgencias de Pediatría y su impacto en la toma de decisiones terapéuticas.
Material y métodos: estudio descriptivo retrospectivo realizado mediante revisión de resultados microbiológicos e historias clínicas electrónicas de los niños de 0 a 15 años atendidos en el periodo de un año.
Resultados: se solicitaron 921 pruebas correspondientes a 837 pacientes, mayoritariamente urocultivos (416), hemocultivos (175) y coprocultivos (136). El laboratorio informó 246 microorganismos, fundamentalmente bacterias (91%). Se prescribieron antimicrobianos a 333 pacientes. Tras la revisión de los resultados microbiológicos, se modificó el tratamiento en 109 pacientes (13%): 96 interrupciones del antimicrobiano prescrito, tres cambios y diez prescripciones nuevas. Se notificaron los resultados a 381 pacientes, 218 del grupo de los resultados positivos y 163 en el de los negativos. El urocultivo fue la muestra implicada en el 63% de las modificaciones terapéuticas.
Conclusiones: el seguimiento de las pruebas microbiológicas de los pacientes que acuden a Urgencias y la modificación de los tratamientos antimicrobianos prescritos pueden ser herramientas útiles en la mejora del uso de antimicrobianos en los Servicios de Urgencias Pediátricas.
Palabras clave
● Laboratorio ● Microbiología ● UrgenciasINTRODUCCIÓN
Los Servicios de Urgencias de Pediatría (SUP) reciben un número creciente de pacientes sin que se haya constatado un aumento en la población pediátrica que lo justifique. La fiebre y otros síntomas asociados a procesos infecciosos son el motivo de consulta más frecuente1. Si en el proceso diagnóstico se solicitan pruebas al Laboratorio de Microbiología, el resultado suele recibirse cuando el paciente ha abandonado ya el SUP. Esto hace que en raras ocasiones se disponga de la información necesaria para conocer el agente etiológico en el momento del alta, por lo que la prescripción del tratamiento antimicrobiano se establece casi siempre de manera empírica. Serán los resultados del Laboratorio de Microbiología los que confirmarán o no la idoneidad del tratamiento antimicrobiano prescrito, para lo cual es imprescindible hacer un seguimiento de los mismos2.
La subsiguiente modificación del tratamiento antimicrobiano cuando sea preciso jugará un papel fundamental en la disminución del uso inadecuado de antimicrobianos y el control del gasto sanitario innecesario, así como en la prevención del aumento de resistencias bacterianas a los antimicrobianos disponibles3.
El objetivo de este estudio es analizar el seguimiento de los resultados obtenidos del laboratorio de microbiología en un SUP y su influencia en la toma de decisiones terapéuticas.
PACIENTES Y MÉTODOS
Estudio descriptivo retrospectivo de los resultados de las pruebas microbiológicas realizadas a los niños de 0 a 15 años atendidos en el SUP de una clínica urbana en el periodo de un año (de mayo de 2014 a abril de 2015), y de las modificaciones terapéuticas posteriores que se llevaron a cabo tras su evaluación.
El protocolo de seguimiento de resultados de Microbiología consistió en la recogida de los datos del paciente, la fecha y la muestra clínica procesada en un libro de registro. Las muestras se procesaron siguiendo los protocolos habituales de toma y transporte de muestras microbiológicas4.
Los pediatras del equipo del SUP revisaron diariamente los resultados microbiológicos recibidos y los analizaron en el contexto de la historia clínica del paciente ya que, a pesar de que sería lo deseable, los pediatras de la red de Atención Primaria no disponen del acceso necesario. Se contactó vía telefónica con el paciente en los casos de resultado positivo en alguna prueba microbiológica, necesidad de modificar o instaurar tratamientos antimicrobianos, necesidad de control clínico del paciente a tenor de la historia clínica o del microorganismo aislado y por solicitud expresa de los familiares del paciente o su médico de cabecera.
Se consideraron resultados positivos aquellos en los que se aisló o detectó la presencia de microorganismos responsables del cuadro clínico. Se consideraron resultados negativos las contaminaciones y aquellos en los que no se detectaron microorganismos.
Se modificaron aquellos tratamientos que no se correspondieron con los más adecuados según el consenso de tratamiento antimicrobiano empírico de las infecciones en la infancia de la Sociedad de Urgencias de Pediatría (SEUP) y la Sociedad de Infectología Pediátrica (SEIP)5.
Para la realización del estudio se obtuvo permiso del Comité de Ética e Investigación del centro asistencial. Se obtuvo consentimiento informado de los participantes o de sus representantes legales.
El análisis de los datos se realizó con el programa informático IBM SPSS Statisticsz® 20.0.
RESULTADOS
Durante el año de estudio se atendieron 20 813 pacientes y se realizaron 921 pruebas microbiológicas correspondientes a 837 pacientes distintos. El diagnóstico más frecuente fue el síndrome febril sin foco (FSF), en el 22,8% de los casos, seguido de la infección del tracto urinario (ITU) en el 22,5% y la gastroenteritis aguda (GEA), en 14,7%. Las pruebas más solicitadas fueron urocultivo, hemocultivo y coprocultivo (Tabla 1).
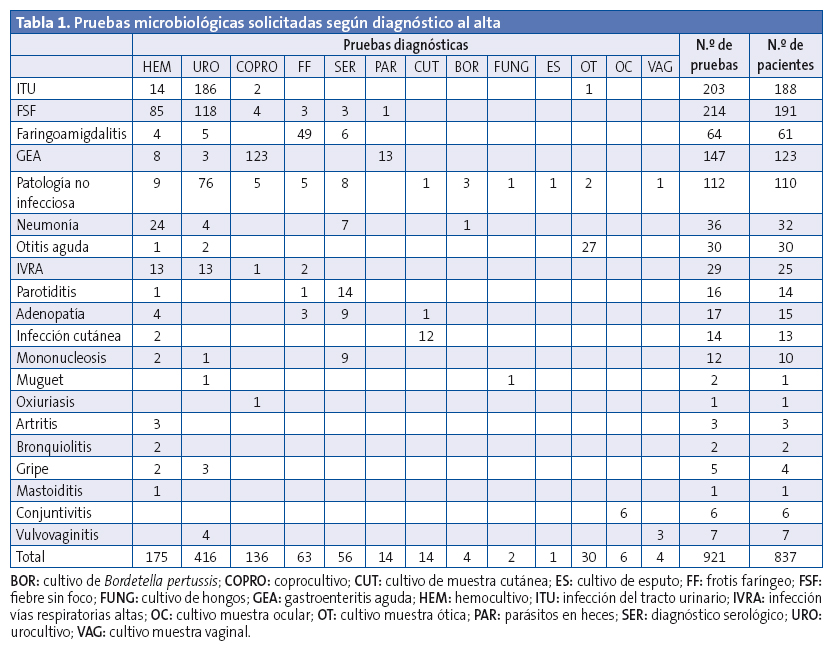
Se obtuvieron 246 resultados de aislamientos positivos: 225 bacterias, 18 virus, dos hongos y un oxiuro. Por microorganismos, E. coli fue el más frecuente (81) seguido de C. jejuni (32), S. pyogenes (22) y S. enterica (19). El 43,7% (98) de los urocultivos fueron positivos. En 22 hemocultivos (12,5%) hubo crecimiento bacteriano, de los que 18 fueron S. epidermidis que, tras revisar la historia clínica, se consideraron contaminantes. Los cuatro restantes correspondieron a E. coli en una ITU con bacteriemia y S. aureus, S. agalactiae y S. pneumoniae en el curso de FSF.
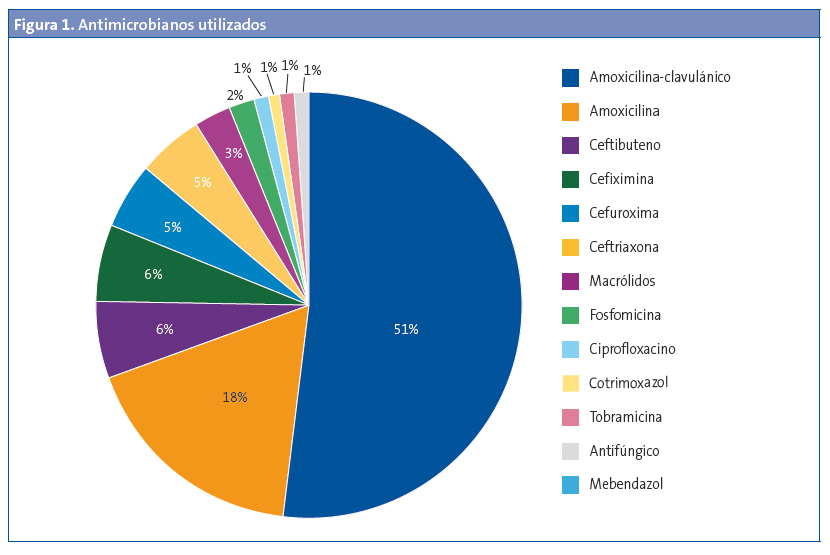
Se prescribieron antimicrobianos de forma empírica a 333 pacientes (Fig. 1).
Se notificó el resultado a 381 pacientes, 218 con resultados positivos y 163 con resultados negativos. El tiempo medio desde la solicitud de la prueba hasta la notificación del resultado fue de 2,7 días, con diferencias según el tipo de prueba realizada (el 65,5% de los urocultivos en menos de 48 horas y el 73% de los coprocultivos pasadas las 48 horas).
Se realizaron intervenciones terapéuticas en 15 pacientes del grupo con resultados positivos: se interrumpieron dos tratamientos antimicrobianos que se consideraron innecesarios (uno con IgM positiva a virus de Epstein-Barr y el otro un aislamiento de S. entérica en coprocultivo), se modificó el tratamiento antimicrobiano elegido en tres casos (amoxicilina por amoxicilina-clavulánico tras aislar S. aureus en el cultivo ótico en dos pacientes y ciprofloxacino por amoxicilina en un paciente con otitis aguda) y se prescribieron tratamientos antimicrobianos a diez pacientes que no habían recibido tratamiento (tres ITU por E. coli, tres GEA por C. jejuni con clínica persistente, un caso de oxiuros, y tres faringoamigdalitis por S. pyogenes). Además, se realizó control clínico en seis pacientes de este grupo: tres se remitieron al SUP (una ITU en tratamiento con cefixima y bacteriemia por E. coli y las bacteriemias por S. pneumoniae y S. agalactiae que se encontraron) y tres se derivaron al pediatra de Atención Primaria (un paciente con E. coli en urocultivo, un paciente con GEA por C. jejuni y un paciente con faringoamigdalitis por S. pyogenes). No se contactó con 28 pacientes del grupo de resultados positivos (11,3%), aunque consta la recepción del informe del laboratorio y su registro en la historia clínica.
Se notificaron 163 resultados negativos por petición expresa de la familia o su médico de cabecera (69 pacientes) o para interrumpir el tratamiento antimicrobiano pautado (94 pacientes).
En total se modificó el tratamiento empírico de 109 pacientes, 15 del grupo de los resultados positivos y 94 en el de los negativos. El urocultivo fue la muestra que suscitó mayor número de modificaciones terapéuticas (63%): 66 urocultivos negativos en pacientes con tratamiento antimicrobiano empírico innecesario y tres urocultivos positivos en pacientes sin tratamiento al alta.
DISCUSIÓN
Se solicitaron pruebas microbiológicas a un número considerable de pacientes durante el año de estudio, el 4% del total de visitas en el SUP. Esto pone de manifiesto la importante carga de trabajo no asistencial que implica el seguimiento de los resultados del Laboratorio de Microbiología en el SUP, para el que habitualmente no se dispone de personal responsable.
La solicitud de test microbiológicos fue motivada fundamentalmente por síndromes febriles en el contexto de procesos infecciosos no filiados y sospechas de ITU o GEA, que fueron los motivos de consulta más frecuentes, como se ha descrito en estudios previos6. Sin embargo, la realización de pruebas microbiológicas no siempre se ajustó a los protocolos diagnóstico-terapéuticos utilizados en Pediatría7, sobre todo en el caso del urocultivo, que se solicitó prácticamente por cualquier tipo de síndrome clínico infeccioso (Tabla 1).
El porcentaje global de resultados positivos de las pruebas microbiológicas solicitadas fue del 26,7%. Se aislaron microorganismos patógenos en el 2,2% de los hemocultivos, porcentaje similar al descrito en otros estudios realizados tras la introducción de la vacuna antineumocócica conjugada8. El diagnóstico de bacteriemia es indicación de revisión pormenorizada del caso y valoración clínica precoz del paciente. En nuestro estudio se realizó control clínico y analítico de los tres pacientes con bacteriemia por E. coli, S. pneumoniae y S. agalactiae, que evolucionaron bien. Sin embargo, no aparecen registros sobre notificación del resultado ni otro tipo de contacto con el paciente que presentó bacteriemia por S. aureus, lo que se considera un error grave de registro y de manejo clínico. En cuanto a los urocultivos, permitieron confirmar la ITU en el 23,5% de los casos en los que se sospechaba, como se ha descrito en otros trabajos9. También fueron útiles para descartar 66 sospechas de ITU no confirmadas e interrumpir el tratamiento antimicrobiano que se había prescrito. Se trata de un porcentaje elevado de falsos positivos para el sedimento urinario (35%), a pesar de existir protocolos bien definidos acerca de los métodos de recogida de orina más adecuados en niños con y sin control de esfínteres4.
Se notificaron los resultados al 45,5% de los pacientes. En el 18% de las ocasiones se trataba de resultados negativos comunicados por petición expresa de los familiares que requerían una “garantía de salud” del paciente y no supusieron ninguna ventaja para el proceso de revisión de resultados. A la inversa, no consta la notificación de 28 resultados positivos, lo que se considera inadecuado.
En conclusión, el seguimiento de las pruebas microbiológicas de los pacientes que acudieron al SUP fue útil, ya que permitió modificar el 32,7% de los tratamientos. Por otro lado, se suspendieron todos los tratamientos antimicrobianos innecesarios y se prescribieron aquellos necesarios en los casos con resultados positivos que lo precisaron. Además, el análisis de todo el proceso permitió identificar áreas de mejora que podrían contribuir a la mejora del uso de tratamiento antimicrobiano en el SUP.
Los autores son conscientes de las importantes limitaciones del estudio. El carácter retrospectivo del mismo puede haber originado pérdidas de información no reflejada en el informe de alta o en el libro de registro de resultados. La revisión del cuadro clínico descrito en los informes de alta, sobre todo en aquellos más escuetos, puede haber sufrido interpretaciones subjetivas por parte del revisor. Por último, el diagnóstico al alta, del cual depende la calidad de la indicación del tratamiento antimicrobiano, se ha dado por certero sin que exista la posibilidad de verificarlo.
CONFLICTO DE INTERESES
Las autoras declaran no presentar conflictos de intereses en relación con la preparación y publicación de este artículo.
ABREVIATURAS: FSF: síndrome febril sin foco · GEA: gastroenteritis aguda · ITU: infección del tracto urinario · SEIP: Sociedad de Infectología Pediátrica · SEUP: Sociedad de Urgencias de Pediatría · SUP: Servicios de Urgencias de Pediatría.
BIBLIOGRAFÍA
- Schneider SM, Gallery ME, Schafermeyer R, Zwemer FL. Emergency department crowding: a point in time. Ann Emerg Med. 2003;42:167-72.
- May L, Cosgrove S, L’Archeveque M, Talan DA, Payne P, Jordan J, et al. A call to action for antimicrobial stewardship in the emergency department: approaches and strategies. Ann Emerg Med. 2013;62:69-70.
- Dellit TH, Owens RC, McGowan JE, Gerding DN, Weinstein RA, Burke JP, et al. Infectious Diseases Society of America and the Society for Healthcare Epidemiology of America guidelines for developing an institutional program to enhance antimicrobial stewardship. Clin Infect Dis. 2007;44:159-77.
- Servicio de Microbiología del Hospital Donostia.Protocolo de toma y transporte de muestras para Microbiología. En: Osakidetza [en línea] [consultado el 31/05/2017]. Disponible en www.osakidetza.euskadi.eus/contenidos/informacion/hd_publicaciones/es_hdon/adjuntos/Protocolo42MuestrasMicrobiologia.pdf
- De la Torre M, Pociello N, Rojo P, Saavedra J. Tratamiento antimicrobiano empírico de las infecciones en la infancia. Consenso SEUP/SEIP. En: SEUP [en línea] [consultado el 31/05/2017]. Disponible en www.seup.org/pdf_public/pub/tto_antimicrobiano.pdf
- Mintegi S, Benito J, García S, Corrales A, Bartolomé M, Trebolazabala N. Demanda y asistencia en un servicio de urgencias hospitalario. An Pediatr (Barc). 2004;61:156-61.
- Asociación Española de Pediatría, Sociedad Española de Infectología Pediátrica. Protocolos de Infectología. En: Asociación Española de Pediatría [en línea] [consultado el 31/05/2017]. Disponible en www.aeped.es/documentos/protocolos-infectologia
- Rodríguez Fanjul J, Hernández-Bou S, Trenchs Sainz de la Maza V, Luaces Cubells C. Estudio descriptivo de los hemocultivos positivos en un servicio de urgencias pediátrico. Emergencias. 2012;24:386-8.
- Jiménez Carrascosa M, Ignacio Cerro C, Míguez Navarro C, Acero Rivas O, Pascual Fernández C, Cañadas Franco V. Cultivos de orina recogidos en un Servicio de Urgencias. En: Sociedad Española de Enfermería de Urgencias y Emergencias [en línea] [consultado el 31/05/2017]. Disponible en http://enfermeriadeurgencias.com/ciber/PRIMERA_EPOCA/2007/octubre/cultivosdeorina.htm




