Vol. 15 - Num. 58
Originales
Estudio sobre la eficacia y utilidad de la solución salina hipertónica al 3% en la bronquiolitis aguda del lactante hospitalizado
Raquel Martín Martína, G Yep Chullenb, Marciano Sánchez Baylec, Enrique Villalobos Pintod, Patricia Flores Pérezd
aPediatra. CS Párroco Julio Morate. Madrid. España.
bServicio de Pediatría, Hospital Infantil del Niño Jesús. Madrid. España.
cPediatra. Fundación para la Investigación, Estudio y Desarrollo de la Salud Pública. Madrid. España.
dSección de Pediatría Hospitalaria. Hospital Infantil Universitario Niño Jesús. Madrid. España.
Correspondencia: R Martín. Correo electrónico: raquelmartin333@hotmail.com
Cómo citar este artículo: Martín Martín R, Yep Chullen G, Sánchez Bayle M, Villalobos Pinto E, Flores Pérez P. Estudio sobre la eficacia y utilidad de la solución salina hipertónica al 3% en la bronquiolitis aguda del lactante hospitalizado. Rev Pediatr Aten Primaria. 2013;15:109-15.
Publicado en Internet: 20-06-2013 - Número de visitas: 46558
Resumen
Objetivo: estudiar la utilidad de la solución salina hipertónica (SSH) al 3% inhalada en el tratamiento de la bronquiolitis aguda (BA) del lactante hospitalizado.
Pacientes y métodos: estudio de casos y controles realizado con 639 pacientes de edad inferior a siete meses e ingresados con diagnóstico de BA, primer episodio, durante tres periodos estacionales consecutivos, en la sección de lactantes de un hospital pediátrico de Madrid (España). Los pacientes que recibieron como tratamiento, durante los dos primeros periodos estacionales, suero salino fisiológico (SSF) inhalado con o sin medicación se consideraron el grupo control y los pacientes que recibieron, durante el tercer periodo estacional, suero salino hipertónico al 3% inhalado con o sin medicación se consideraron como casos. Los días de hospitalización y las horas de oxigenoterapia fueron utilizados como medidas de resultado.
Resultados: de la totalidad de los niños estudiados, 460 recibieron SSF inhalado, y 179 recibieron SSH al 3%. En el grupo que recibió SSF, la estancia media en el hospital fue de 5,16 días (intervalo de confianza del 95% [IC 95%]: 4,78-5,56) y el tiempo medio de oxigenoterapia fue de 57,34 (IC 95%: 52,93-61,75) frente a 4,90 días (IC 95%: 4,64-5,07) y 67,53 horas (IC 95%: 60,36-74,69), respectivamente, en el grupo tratado con SSH. Estos resultados no alcanzan significación estadística. Los pacientes con estudio positivo de virus respiratorio sincitial (VRS) en aspirado nasofaríngeo y que recibieron SSF necesitaron menos horas de oxígeno de manera significativa (p=0,004), así como aquellos que tenían edad <3 meses (p=0,007).
Conclusiones: los resultados obtenidos muestran que la SSH al 3% inhalada no resulta eficaz para reducir la estancia hospitalaria ni el tiempo de oxigenoterapia en los pacientes con BA; además, en los niños menores de tres meses y con estudio positivo de VRS en aspirado nasofaríngeo la aplicación de SSF inhalado consiguió una necesidad menor de horas de oxígeno.
Palabras clave
● Bronquiolitis aguda ● TratamientoINTRODUCCIÓN
La bronquiolitis aguda (BA) es la infección de las vías respiratorias bajas más frecuente en los niños menores de un año, siendo los lactantes más pequeños los que precisan hospitalización con más frecuencia y se ven sometidos a intervenciones terapéuticas y pruebas diagnósticas cuya eficacia y utilidad no están suficientemente demostradas1.
Probablemente, la BA sea una de las patologías pediátricas más estudiadas, son numerosas las guías de práctica clínica y las recomendaciones de grupos de expertos2,3 y a pesar de la información publicada no hay consenso a la hora de aplicar el tratamiento a estos pacientes. El desajuste entre la práctica clínica y la evidencia científica genera un elevado e injustificado consumo de recursos sociales y económicos4,5.
La BA se caracteriza por una inflamación aguda de los bronquiolos terminales, siendo el edema de las vías respiratorias y el taponamiento por moco las características anatomopatológicas predominantes, es por eso que cualquier modalidad terapéutica que pueda reducir estas alteraciones y mejorar la eliminación de las secreciones puede ser beneficiosa6.
Las soluciones salinas hipertónicas (SSH) están compuestas por cloruro de sodio disuelto en agua destilada, concretamente la elaboración de suero salino al 3% se realiza con la mezcla de suero salino fisiológico (SSF) y cloruro sódico al 20%7 y su uso en forma de inhalaciones ha demostrado mejorar el aclaramiento mucociliar in vivo e in vitro en enfermedades como la fibrosis quística, el asma y las bronquiectasias. La oxigenoterapia ha sido el único tratamiento capaz de mejorar el curso clínico de la BA, por eso el manejo de estos pacientes se basa en las medidas generales de soporte8.
El objetivo del presente trabajo es estudiar la eficacia de las inhalaciones de SSH al 3%, aplicadas en el tratamiento del primer episodio de bronquiolitis aguda a un grupo de lactantes hospitalizados en el Hospital Infantil del Niño Jesús de Madrid.
PACIENTES Y MÉTODOS
Se trata de un estudio de casos y controles, realizado con 639 pacientes de edad inferior a siete meses y que ingresaron con diagnóstico de BA, primer episodio, en la sección de lactantes del Hospital Infantil del Niño Jesús de Madrid, durante los meses de invierno de los años 2007-2008, 2008-2009 y 2009-2010.
No se realizó el cálculo del tamaño muestral por considerar que el número habitual de ingresos con el diagnóstico de BA en el periodo seleccionado era el adecuado y por la imprevisibilidad de la incidencia de la enfermedad.
Se diagnosticó BA a los niños con historia previa de infección respiratoria de vías altas y clínica de distrés respiratorio y auscultación cardiopulmonar con sibilancias o sibilancias y roncus (criterios de McConochie). Fueron ingresados los niños que presentaron al menos uno de los siguientes signos en la consulta de la urgencia hospitalaria: aspecto tóxico, historia de apnea/cianosis, frecuencia respiratoria/minuto >60 y saturación de oxígeno <94%.
Fueron excluidos del estudio los niños con problemas respiratorios crónicos, cardiopatías, y aquellos con BA cuya gravedad inicial necesitó ingreso en la unidad de Cuidados Intensivos.
Para realizar el estudio y durante los periodos estacionales de 2007-2008 y 2008-2009 los pacientes recibieron SSF inhalado con o sin medicación, considerando a este grupo como controles; durante el periodo estacional de 2009-2010, los pacientes recibieron SSH al 3% con o sin medicación y se consideró a estos pacientes como casos.
Las intervenciones consistieron en la administración de 3 cc de suero salino aplicado con un nebulizador estándar con oxígeno cada ocho horas si estaban sin medicación y cada cuatro o seis horas si tenían medicación.
Las medidas de resultado utilizadas en este trabajo han sido los días de hospitalización y las horas de oxigenoterapia recibidas.
El criterio para cesar la oxigenoterapia fue obtener cifras de saturación de oxígeno =94%. La saturación de oxígeno era registrada por la enfermería cada cuatro horas.
Los criterios de alta hospitalaria fueron estar afebril, con buen estado general, tolerar alimentación oral y no precisar oxígeno.
El análisis estadístico se realizó con el programa comercial SPSS®11.0. Los datos básicos se expresaron en medias y desviaciones estándar en el caso de las variables cuantitativas y en números y porcentaje en el caso de las variables cualitativas. Se calcularon los intervalos de confianza del 95% (IC 95%).
Las comparaciones entre las variables cuantitativas se realizaron mediante el test de Mann-Whitney después de comprobar que no se ajustaban a una distribución normal (test de Kolmogorov-Smirnov). En las que se realizaron entre las variables cualitativas se utilizó la prueba de chi al cuadrado. Se consideró significación estadística para p valores inferiores a 0,05.
RESULTADOS
El total de pacientes ingresados con diagnóstico de BA y edad inferior a siete meses durante los periodos estacionales de 2007-2008, 2008-2009, 2009-2010 que se incluyeron en el estudio fueron 639, de los cuales 237 (37,08%) eran niños y el resto niñas. Durante los dos primeros periodos estacionales 460 pacientes recibieron tratamiento con SSF inhalado con o sin medicación broncodilatadora, constituyendo el grupo control; durante el último periodo estacional 179 pacientes recibieron SSH al 3% solo o con medicación broncodilatadora y formaron el grupo de casos.
El rango de edad de los pacientes fue de diez días a 6,5 meses y teniendo en cuenta que los más pequeños sufren los episodios de BA más graves se hizo una distribución según la edad en <3 meses y =3 meses.
La Tabla 1 muestra las características generales de los pacientes y la comparación en los dos grupos según el tratamiento recibido, observándose que no hay diferencia significativa entre ellos.
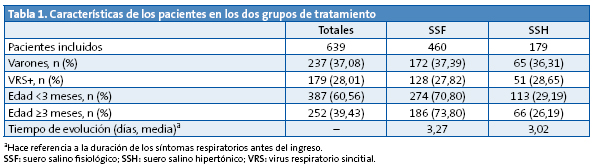
La Tabla 2 refleja los resultados evolutivos del conjunto de los pacientes estudiados según el tratamiento recibido y no se ha encontrado diferencia significativa en cuanto a los días de ingreso y sí en cuanto al menor número de horas de oxigenoterapia que precisó el grupo tratado con SSF (p=0,001).
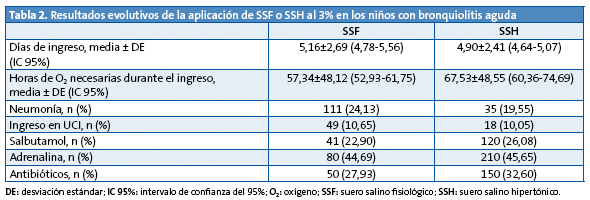
En la Tabla 3 aparecen los resultados obtenidos en relación al hallazgo de virus respiratorio sincitial (VRS) positivo o negativo en el aspirado nasofaríngeo y no se ha encontrado diferencia significativa en cuanto a los días de ingreso, pero sí en cuanto al menor número de horas de oxigenoterapia en los pacientes VRS+ tratados con SSF (p=0,004).
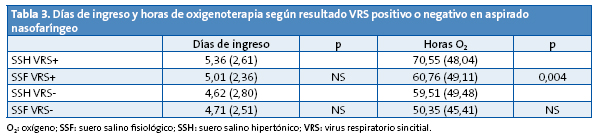
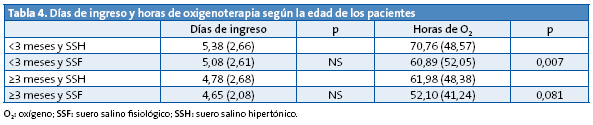
La Tabla 4 analiza los resultados según que la edad de los pacientes fuera menor o igual/mayor de tres meses; en este caso se encuentra, de manera significativa, una menor necesidad de oxigenoterapia en los <3 meses tratados con SSF (p=0,007). En los de =3 meses y en relación al número de horas de oxigenoterapia, la significación estadística del resultado obtenido está en el límite (p=0,081).
DISCUSIÓN
Este trabajo aporta la evaluación del tratamiento con SSH al 3% inhalado, administrado a lactantes hospitalizados por un primer episodio de BA y utilizando para valorar los resultados obtenidos el tiempo de hospitalización y las horas de oxigenoterapia empleadas.
Desde hace tiempo se conoce que las SSH aumentan la depuración mucociliar en pacientes normales9 y que son útiles y seguras como estrategia terapéutica en enfermedades con aclaramiento mucociliar defectuoso: asma, bronquiectasias y mucoviscidosis10,11.
Los potenciales efectos beneficiosos del suero salino hipertónico pueden deberse a la capacidad teórica para disminuir la viscosidad y elasticidad del gel de moco, rompiendo los enlaces iónicos dentro del mismo; provocar un flujo osmótico de agua hacia el interior de la capa de moco, con lo cual mejora su reología y se rehidrata; estimular la movilidad ciliar mediante la liberación de prostaglandinas; reducir el edema de la pared al absorber agua de la mucosa y submucosa o, por último, inducir el esputo y la tos12.
La bibliografía consultada muestra estudios realizados con pacientes hospitalizados y trabajos con pacientes que han acudido a los Servicios de Urgencias y no han llegado a ingresar. La selección de variables de resultado son diferentes en cada caso y esto hace que los resultados obtenidos en los pacientes hospitalizados no sean extrapolables a los ambulatorios y viceversa, lo cual supone un sesgo en cada caso y por supuesto en el nuestro.
Por lo que respecta al tiempo de hospitalización, la estancia media por BA en la bibliografía es de 3-5 días y nuestros resultados coinciden en ese aspecto con una media de 4,9 días en los niños que han recibido SSH y de 5,16 días los que recibieron SSF. La administración de SSH al 3% en nuestro caso no ha logrado reducir ese periodo de manera significativa; la aplicación de SSF tampoco obtuvo beneficios en este sentido. La revisión Cochrane consultada13 presenta tres estudios hospitalarios14-16 donde los autores muestran resultados estadísticamente significativos, con reducción de 0,9 días en la estancia hospitalaria, tras administrar SSH al 3%; sin embargo, es obvio que la magnitud clínica del resultado es irrelevante. La propia revisión Cochrane ha sido objeto de evaluación crítica por otros autores17-20, ya que establecer una función terapéutica para la SSH tiene implicaciones clínicas importantes. Luo Z, et al.21,22 en sus trabajos concluyen que la aplicación de SSH al 3% es segura y eficaz, reduciendo de manera significativa la estancia hospitalaria de los pacientes estudiados.
Los niños incluidos en este trabajo han precisado oxigenoterapia durante su hospitalización y las horas que han necesitado han sido otra medida de resultado. Hemos comprobado que las necesidades de oxigenoterapia se han visto reducidas significativamente en el grupo de niños menores de tres meses a los que se aplicó SSF inhalado; además, los niños con resultado analítico VRS+ en aspirado nasofaríngeo y que recibieron aerosoles de SSF también necesitaron menos horas de oxigenoterapia. La SSH al 3% no mostró beneficio en ningún caso. Es necesario resaltar que estos resultados no son extrapolables a los pacientes ambulatorios y que, por tanto, no han precisado de oxigenoterapia. Hemos analizado separadamente el grupo con VRS+ para evaluar si la eficacia del tratamiento era distinta de acuerdo a la etiología de la BA. La explicación de estos resultados, relacionados con la edad y con un resultado VRS+, la desconocemos y no hemos encontrado otras referencias en la bibliografía consultada.
En los estudios realizados con pacientes ambulatorios, las medidas de resultado para valorar la eficacia del tratamiento consisten en valorar la mejoría de los síntomas tras la aplicación del mismo y en cuantificar la disminución del porcentaje de ingresos. La mayoría de los trabajos consultados23-25 no han encontrado diferencia significativa entre la aplicación de SSF y SSH al 3%; Al-Ansari, et al.26 han evaluado soluciones salinas al 5, 3 y 0,9% y han encontrado más eficaz la solución al 5%. La revisión Cochrane recoge un trabajo27 realizado con pacientes ambulatorios y cuyos autores han encontrado resultados significativamente beneficiosos al administrar SSH al 3%.
No hemos registrado efectos adversos al aplicar el tratamiento y, en este sentido, todos los autores coinciden al asegurar que es inocuo, seguro y económico aplicar el suero salino al 3% solo28 o con broncodilatadores.
Una limitación de nuestro trabajo es que no se ha realizado una asignación aleatoria al tratamiento de los pacientes.
Como conclusión, destacamos que la SSH al 3% con medicación o sin ella administrada a los pacientes ingresados en nuestro hospital por BA no ha resultado eficaz para reducir la estancia hospitalaria o el tiempo de oxigenoterapia. Teniendo en cuenta la prevalencia de la BA, su repercusión a nivel social y económico, sería deseable insistir en futuros estudios sobre este tema.
CONFLICTO DE INTERESES
El autor declara no presentar conflictos de intereses en relación con la preparación y publicación de este artículo.
ABREVIATURAS: BA: bronquiolitis aguda • IC 95%: intervalo de confianza del 95% • SSF: suero salino fisiológico • SSH: solución salina hipertónica • VRS: virus respiratorio sincitial.
BIBLIOGRAFÍA
- González de Dios J, Ochoa Sangrador C, Grupo Investigador del Proyecto aBREVIADo (BRonquiolitis-Estudio de Variabilidad Idoneidad y Adecuación). Estudio de variabilidad en el abordaje de la bronquiolitis aguda en España en relación con la edad de los pacientes. An Pediatr (Barc). 2010;72:4-18.
- Simó Nebot M, Claret Teruel G, Luaces Cubells C, Estrada Sabadell MD, Pou Fernández J. Guía de práctica clínica sobre la bronquiolitis aguda: recomendaciones para la práctica clínica. An Pediatr (Barc). 2010;73:208.e1-10.
- González de Dios J, Ochoa Sangrador C, Grupo de Trabajo (Grupo Investigador, Grupo de Revisión y Panel de Expertos) del Proyecto aBREVIADo (Bronquiolitis-Estudio de Variabilidad, Idoneidad y Adecuación). Recomendaciones de la Conferencia de Consenso de Bronquiolitis aguda en España: de la evidencia a la práctica. Rev Pediatr Aten Primaria. 2010;12 (Supl 19):s107-128.
- Sánchez Etxaniz J, Benito Fernández J, Mintegi Raso S. Bronquiolitis aguda: ¿por qué no se aplica lo que se publica? Barreras en la transmisión del conocimiento. Rev Pediatr Aten Primaria. 2008;10:23-32.
- Calogero C, Sly PD. Acute viral bronchiolitis: To treat or not to treat. That is the question. J Pediatr. 2007;151(3):235-37.
- Mandelberg A, Amirav I. Hypertonic saline or high volume normal saline for viral bronchiolitis: mechanisms and rationale. Pediatr Pulmonology. 2010;45(1):36-40.
- Borja Urbano G, Pérez Pérez G, Andrés Martín A, Navarro Merino M. Actualización en el manejo de la bronquiolitis. Vox Paediatrica. 2011;XVIII(2):57-67.
- Pérez Rodríguez MJ, Otheo de Tejada Barasoain E, Ros Pérez P. Bronquiolitis en pediatría: puesta al día. Inf Ter Sist Nac Salud. 2010;34:3-11.
- Daviskas E, Anderson SD, Gonda I, Eberl S, Meikle S, Seale P, et al. Inhalation of hypertonic saline aerosol enhances mucociliary clearance in asthmatic and healthy subjects. Eur Respir J. 1996;9:725-32.
- Robinson M, Regnis JA, Bailey DL, King M, Bautovich GJ, Bye PTP. Effect of hypertonic saline, amiloride and cough on muciciliary clearance in patients with cystic fibrosis. Am J Respir Crit Care Med. 1996;153:1503-9.
- Fraga Betancur DA, Baez JC, Reyes JM, Sussini MA. Uso de soluciones salinas hipertónicas inhaladas para restaurar la hidratación superficial de la vía aérea en pacientes con fibrosis quística. Rev Posgrado de la VI.ª Cátedra de Medicina. 2006;164:13-6.
- González de Dios J, Ochoa Sangrador C, Grupo de Revisión del Proyecto aBREVIADo (Bronquiolitis-Estudio de Variabilidad, Idoneidad y Adecuación). Conferencia de Consenso sobre bronquiolitis aguda (IV): tratamiento de la bronquiolitis aguda: Revisión de la evidencia científica. An Pediatr (Barc). 2010;72:285.e1-42.
- Zhang L, Mendoza-Sassi RA, Wainwright C, Klassen TP. Nebulized hypertonic saline solution for acute bronchiolitis in infants. Cochrane Database Syst Rev. 2008;4:CD006458.
- Mandelberg A, Tal G, Witzling M, Someck E, Houri S, Balin A, et al. Nebulized 3% hypertonic saline solution treatment in hospitalized infants with viral bronchiolitis. Chest. 2003;123:481-7.
- Tal G, Cesar K, Oron A, Houri S, Ballin A, Mandelberg A. Hypertonic saline/epinephrine treatment in hospitalized infants with viral bronchiolitis reduces hospitalization stay: 2 years experience. Isr Med Assoc J. 2006;8(3):169-73.
- Kuzik BA, Al-Qadhi SA, Kent S, Flavin MP, Hopman W, Hotte S, et al. Nebulized hypertonic saline in the treatment of viral bronchiolitis in infants. J Pediatr. 2007;151:266-70.
- Fernández Rodríguez M, Martín Muñoz P. Los aerosoles con suero salino hipertónico al 3% podrían disminuir la duración de la hospitalización en lactantes con bronquiolitis. Rev Pediatr Aten Primaria. 2008;10:91-5.
- Balaguer Santamaría A, Buñuel Álvarez JC, González de Dios J. El suero salino hipertónico nebulizado puede disminuir la duración del ingreso hospitalario en lactantes con bronquiolitis aguda. Evid Pediatr. 2009;1:5.
- Horner D, Jenner R. Nebulised hypertonic saline significantly decreases length of hospital stay and reduces symptoms in children with bronchiolitis. Emerg Med J. 2009;26(7):518-9.
- Mathew JL. Hypertonic saline nebulization for bronchiolitis. Indian Pediatrics. 2008;45:987-9.
- Luo Z, Liu E, Luo J, Li S, Zeng F, Yang X, et al. Nebulized hypertonic saline/salbutamol solution treatment in hospitalized children with mild to moderate bronchiolitis. Pediatr Int. 2010;52(2):199-202.
- Luo Z, Fu Z, Liu E, Xu X, Fu X, Peng D, et al. Nebulized hypertonic saline treatment in hospitalized children with moderate to severe viral bronchiolitis. Clin Microbiol Infect. 2011;17(12):1829-33.
- Grewal S, Ali S, McConnell DW, Vandermeer B, Klassen TP. A randomized trial of nebulized 3% hypertonic saline with epinephrine in the treatment of acute bronchiolitis in the emergency department. Arch Pediatr Adolesc Med. 2009;163(11):1007-12.
- Kuzik BA, Flavin MP, Kent S, Zielinski D, Kwan Ch W, Adeleye A, et al. Effect of inhaled hypertonic saline on hospital admission rate in children with viral bronchiolitis: a randomized trial. CJEM. 2010;12(6):477-84.
- Anil AB, Anil M, Saglam AB, Cetin N, Bal A, Aksu N. High volume normal saline alone is as effective as nebulized salbutamol normal saline, epinephrine-normal saline, and 3% saline in mild bronchiolitis. Pediatr Pulmonol. 2010;45(1):41-7.
- Al-Ansari K, Sakran M, Davidson BL, El Sayyed R, Mahjoub H, Ibrahim K. Nebulized 5% or 3% hypertonic or 0,9% saline for treating acute bronchiolitis in infants. J Pediatr. 2010;157(4):630-4.
- Sarrell EM, Tal G, Witzling M, Someck E, Houri S, Cohen HA, et al. Nebulized 3% hypertonic saline solution treatment in ambulatory children with viral bronchiolitis decreases symptoms. Chest. 2002;122(6):2015-20.
- Ralston S, Hill V, Martinez M. Nebulized hypertonic saline without adjunctive bronchodilators for children with bronchiolitis. Pediatrics. 2010;126:e520-e525.
Comentarios
Este artículo aún no tiene comentarios.





